Research conducted by Answers in Genesis staff scientists or sponsored by Answers in Genesis is funded solely by supporters’ donations.
Abstract
Archaeopteryx has been one of the most studied fossils since its discovery in 1861. Yet, the classification of the species is still under debate. Many controversies remain regarding the feather, taxonomic classification, flight capabilities, its “evolution,” and whether Archaeopteryx is a transitional form, a reptile with feathers, or a bird. This paper’s scope is to briefly discuss the latter controversy because it has kept creation and secular scientists’ view divided. The history of Archaeopteryx findings, their historical interpretations, the role of Linnaean and cladistics classification methods, and the implications of all that on Archaeopteryx’s classification were analyzed. This analysis demonstrates its placement as a bird is supported.
This paper also demonstrates that the attempts to place Archaeopteryx’s placement as a dinosaur with feathers rely on evolutionary-based cladistics, which presents many methodological problems. For this reason, it should not be used in the analytical approach for Archaeopteryx, as for any fossil, it is impossible to strip out the evolutionary bias. We conclude that there is no reasonable explanation based on cladistics and its use in baraminological studies to support Archaeopteryx placement other than in the bird group.
Therefore, based on a biblical worldview, logical grounds, the anatomy of the skeleton and skull, the presence of feathers, and following the traditional Linnaean classification, no reason remains for Archaeopteryx to be classified as anything other than a bird.
Introduction
Archaeopteryx lithographica’s skeletal specimens and the feather related to this species were found in the nineteenth century. They have been among the most studied fossils, but there are still many controversial details about them. The first skeletal fossil material related to Archaeopteryx was found two years after one of the publications that has shaped the scientific world the most, Charles Darwin’s book, On the Origin of Species. For some people at that time, the finding of Archaeopteryx seemed to offer a solution to the challenge of missing links or transitional forms, an idea essential to Darwin’s theory of evolution.
In the list of controversies among secular scientists are the quests to determine whether the feather (BSP 1869 VIII 1-main slab, and MB.Av.100-counter slab) belongs to that taxon, whether the specimens of Archaeopteryx represent that genus and only one species, and whether Archaeopteryx would be the first and “youngest” (in an evolutionary perspective) avian. Also yet to be resolved are its flight capabilities and how that ability “evolved,” and lastly, whether Archaeopteryx is a bird, a transitional form, or a dinosaur with feathers—that is, a feathered theropod dinosaur. Regarding that last topic, creation scientists’ views have been divided between two different and opposite conclusions, one group considering Archaeopteryx as a bird and the other as a reptile with feathers—a feathered theropod dinosaur (McLain, Petrone, and Speights 2018). Some supporters of the latter opinion (that Archaeopteryx is a reptile with feathers) seem to no longer consider the classical biological and taxonomic significance of these two different classes—birds and reptiles (McLain, Petrone, and Speights 2018). However, Archaeopteryx’s classification will be the only controversy discussed in this paper.
In the creationist view, Archaeopteryx is a beautiful demonstration of God’s created handiwork—an animal with fully developed feathers, very similar to the modern ones we see in birds today. It is also essential because its fossil record points us to the Genesis Flood. Four skeletons of Archaeopteryx show the neck recurved, which has been demonstrated to happen in water with high salt levels (hypersaline) (Wellnhofer 2009). Secular scientists thus propose its death by drowning (Wellnhofer 2009), which is a reasonable explanation and agrees with the cause of death of many animals during Noah’s Flood. In a catastrophic event such as the Flood of Genesis, we would expect evidence of an elevated salt concentration in the water.
For secular scientists, Archaeopteryx has been considered to play an essential role in understanding the evolution and origin of birds. Therefore, this matter needs to be carefully analyzed since its implications are crucial for secular scientists and creationists.
An Overview of the Known Specimens of Archaeopteryx
The first partial body specimen of Archaeopteryx was discovered in 1855 and described in 1857 by Hermann Meyer. Today it is known as The Haarlem specimen (TM 6428/29). However, at the time, it was classified as a pterosaur. More than 100 years passed until it was described as Archaeopteryx by John Ostrom in 1972 (Ostrom 1972; Wellnhofer 2009). The same specimen was also named with a different genus and species by Wellnhofer in 1970.
Archaeopteryx means ancient feather or ancient wing in Greek. In 1861, a single isolated feather looking just like a modern feather was found in southern Germany, in rocks of the Late Jurassic System, in which all the other known remains of Archaeopteryx were found. This single feather is also known as the first fossil feather ever found. It also represents the first fossil material found and identified as Archaeopteryx. In 1862, Hermann Meyer described and published on the feather, naming it Archaeopteryx lithographica, the name of the animal to which the feather belonged and not the feather itself. He considered the feather to belong to an immature or young individual. Griffiths (1996) concluded that the evidence of the feather (discovered in 1861) and the first skeletal specimens of the animal (the London specimen) is insufficient to place them in the same species. That has still been in dispute. In a press interview, Kaye et al. (2019) stated, based on their research published in the same year, that the feather did not belong to Archaeopteryx (Carney, Tischlinger, and Shawkey 2020). However, Carney, Tischlinger, and Shawkey (2020) concluded that it did belong to Archaeopteryx.
The first correctly identified body specimen of Archaeopteryx was discovered in 1861. It is known as the London specimen housed in the Natural History Museum in London (BMNH 37001). Richard Owen (1862) published his conclusions on Archaeopteryx as a long-tailed bird. Even though he acknowledged its rare peculiarities, Owen considered them an indication of a different order in the Aves Class.
Despite its first formal publication in 1863, the first description of that specimen was presented by Andreas Wagner, a paleontology professor, in 1861 based on the reports of Oppel, his assistant, and Witte, a fossil collector and expert in fossils. Wagner had not seen the fossil before publishing about it. Neither did he have any additional information to support his conclusions. Contrary to Oppel’s opinion that the fossil was a bird with a reptilian tail, Wagner described it as a fossil reptile with feather-like structures covering its body. According to Wagner, only a bird could have feathers, and because of his old-earth creation perspective, a bird could not have existed around that time (in the Jurassic). He also went against Witte, who saw in the fossil features of a bird and a reptile. In the same year (1861), Meyer, who had discovered the feather, stated that the London specimen showed differences from living birds (Wellnhofer 2009).
In a letter dated 1862 to the British Museum, a physician and fossil collector, Dr. Karl Haberlein, described Archaeopteryx as having a tail “differently formed as in a pterodactyl.” In 1863, a zoology professor at Halle University named Giebel declared it was a fake fossil.
| Hermann Meyer | 1857 | A Pterodactylus |
| Friedrich Witte | 1861 | Features of bird and reptile |
| Albert Oppel | 1861 | A bird with a reptilian tail |
| Andreas Wagner | 1861 | A reptile with a feather-like body cover |
| Hermann Meyer | 1861 | It showed differences from living birds |
| Karl Häberlein | 1862 | It has a tail like a pterodactyl |
| C. Giebel | 1863 | An artifact, fake |
| Richard Owen | 1863 | A long-tailed bird |
Table 1 summarizes early investigators’ views about the first Archaeopteryx specimens.
From 1855 to 2014, one isolated feather and 12 Archaeopteryx specimens were discovered and announced to the public. Formal descriptions were published in the years following their finds. They are mainly named based on the order in which they were discovered or where they are housed. The skeletal specimens of Archaeopteryx lithographica have also been named differently by scientists who concluded that some specimens do not represent this taxon. Those names are listed in the following list of Archaeopteryx material:
- The feather (BSP 1869 VIII 1 and MB.Av.100)
- The London specimen (BMNH 37001)
- Griphosaurus Wagner (1862)
- Griphosaurus problematicus Woodward (1862)
- Griphornis longicaudatus Woodward (1862)
- Archeopteryx macrurus Owen (1863)
- Archaeopteryx macrura Owen (1863)
- Archaeopteryx oweni Petronievics (1921)
- The Berlin specimen (HMN 1880/1881)
- Archaeopteryx macrura Owen (1863)—Dames (1884)
- Archaeopteryx siemensii Dames (1897)—Elżanowski (2002); Mayr et al. (2007)
- Archaeornis siemensii Dames (1897)—Heilmann (1926); Lambrecht (1933); Petronievics (1921); Stephan (1987)
- The Maxberg specimen (missing today)
- Archaeopteryx sp. Elżanowski (2002)
- The Haarlem specimen (6928 and 6929)
- Pterodactylus crassipes H. v. Meyer (1857, 1859)
- Scaphognathus sp. Wellnhofer (1970)
- Archaeopteryx sp. Elżanowski (2002)
- Ostromia crassipes (an anchiornithid) Foth and Rauhut (2017)
- The Eichstätt specimen (JM 2257)
- Archaeopteryx ? n. sp. Mayr (1973)
- Archaeopteryx recurva Howgate (1984)
- Jurapteryx recurva Howgate (1985); Stephan (1987)
- Archaeopteryx sp. Elżanowski (2002)
- The Solnhofen specimen (BMMS 500)
- Wellnhoferia grandis Elżanowski (2001)
- The Munich specimen (BSP 1999 | 50)
- Archaeopteryx bavarica Wellnhofer (1993); Elżanowski (2002)
- The seventh skeleton of Archaeopteryx Elżanowski and Wellnhofer (1996)
- Archaeopteryx siemensii Dames (1927); Mayr et al. (2007)
- The Eight specimen (SNSB BSPG VN-2010/1)
- Archaeopteryx Mäuser (1997); Tischlinger and Scharf (1998)
- Archaeopteryx sp. Frickhinger (1999)
- Archaeopteryx albersdoerferi Kundrát et al. (2019)
- The Ninth specimen (private ownership)
- Urvogel, Archaeopteryx Röper (2004)
- The Thermopolis specimen (WDC-CSG-100)
- Archaeopterygidae Mayr (2005); Mayr, Pohl, and Peters (2005)
- Archaeopteryx siemensii Dames (1897); Mayr et al. (2007)
- The Eleventh specimen (private ownership)
- Archaeopteryx Foth, Tischlinger, and Rauhut (2014)
- The Twelfth specimen (DNWK 02924)
- Archaeopteryx Rauhut, Foth, and Tischlinger (2018)
Unlike modern birds, Archaeopteryx has some features that Thomas Huxley saw as similarities with dinosaurs. He was the first to propose the relationship between dinosaurs and birds (Huxley 1868). Almost 100 years after Huxley’s proposition, in 1969, Ostrom published (Ostrom 1969) his research on the fossil material found during two expeditions, one in 1931–1932 and the other over 30 years later. In his conclusions, the new species he described, Deinonychus antirrhopus, was a theropod dinosaur (Dromaeosauridae) and shared many similarities with Archaeopteryx. Because of the study of Deinonychus, Ostrom (1976) proposed that birds were descendants of theropods and noted similarities between Archaeopteryx and coelurosaurs. Those were later described in 1991 and 1994 (Ostrom 1991; 1994).
One of the views that have helped to shape the idea of the relationship between Archaeopteryx and dinosaurs was propagated by Gerhard Heilmann, author of one of the books that greatly influenced the discussion of the evolution of birds (Wellnhofer 2009). He was a painter and an illustrator and published his book in 1926. He stated that Archaeopteryx might be termed a “warm-blooded reptile disguised as a bird” (Heilmann 1926, 32).
Following is the list of features that Archaeopteryx has and which are interpreted as shared with dinosaurs. Of course, it is impossible to discuss all of them since that is not the scope of this paper. However, this brief explanation presents principles from which those characteristics can be seen, analyzed, and understood.
The list from Wellnhofer (2009) includes the presence of:
- (1) teeth in its jaws,
- (2) three clawed digits,
- (3) a long bony tail (pygostyle),
- (4) gastralia,
- (5) tetraradiate palatine (Mayr, Pohl, and Peters 2005),
- (6) a hyperextendable claw on the second toe,
- (7) a reduced fifth toe, and
- (8) interdental plates.
Some of those anatomical characteristics can be easily demonstrated to be shared with other birds, such as the presence of teeth in enantiornithines (extinct birds), claws in hoatzins (modern birds), and a bony tail (pygostyle) that is also seen in enantiornithines.
Gastralia are shared with other groups of animals. They are a common trait in tetrapods but are only present in some living reptiles (Claessens 2004).
Other features, such as the palatine, have different but opposite interpretations. Mayr, Pohl, and Peters (2005) state it as tetraradiate (with four processes), which is a dinosaur-like feature, but Elżanowski and Wellnhofer (1996) see it as triradiate (three processes), which is a bird-like feature. That is still in dispute.
O’Connor et al. (2022) state that studies have confirmed that the condition of having teeth separated by interdental bone is typically present in most toothed birds, like Sapeornis (Wang et al. 2017) and enantiornithines like Pengornis (O’Connor and Chiappe 2011; Zhou, Clark, and Zhang 2008).
Other features in the above list and others not cited remind us that scientists might not fully understand these features. It seems trite to say, but in all scientific fields and endeavors, scientists do not understand everything, which is why there is a need to keep researching. There were, are, and will still be many situations in which conclusions cannot be immediately drawn. That demonstrates that scientists also have fallible and finite minds in this fallen world as they try to understand the creatures created by the Creator’s perfect, infinite, and creative mind.
From a biblical perspective, similarities are expected to be seen throughout God’s creation because every creature has the same Designer. So, we should perceive the same designs repeated in different animals. Therefore, similarities found among different created kinds are interpreted as features designed for a purpose that, even though we might not fully understand them, are still designed by God for a reason. On the other hand, looked at through evolutionary lenses, those similarities arose from an evolutionary history of ancestry and descent. The point is not whether there are similarities, but how they are interpreted.
The “Reptile Bird” Alive Today!
The hoatzin (Opisthocomus hoazin) is a bird known as a “reptile bird” because it shares features that are defined as reptilian (Parker 1891). It also shares characteristics with mammals (Grajal 1995). It is called an enigmatic creature with an unsure phylogenetic placement. It has claws like a reptile, feathers like birds, and a digestive system like a cow (Sanders 2019). Thus, it has a mosaic of shared features. The platypus is another example of such a mosaic of shared traits.
A Matter of Classifications, Definitions, and Interpretations—Archaeopteryx as a Transitional Form
Thomas Huxley, a supporter of Darwin and his theory of evolution, proposed in 1868 that Archaeopteryx was a transitional form. He compared Archaeopteryx with Compsognathus, Megalosaurus, and Iguanodon and argued Archaeopteryx was a transitional form. Huxley ended his article by stating: “Archaeopteryx is more remote from the boundary-line between birds and reptiles . . .” (Huxley 1868, 248).
This interpretation relied only on an evolutionary perspective—an a priori belief that animals evolved from one kind into another different kind of animal—rather than on observable evidence. Over the years, this idea has been depauperated. The desire to make Archaeopteryx a missing link was only to support the theory of evolution in answering the critics of Darwin’s proposition that missing links must exist (Wellnhofer 2009). Thus, many scientists and people generally still believe Archaeopteryx is a transitional form.
In 1881, Othniel Charles Marsh created the Theropoda suborder (now clade), grouping all known dinosaurs from the Triassic and the carnivorous dinosaurs from the Jurassic and Cretaceous. Jacques Gauthier described theropods in 1986 via cladistics (an evolutionary method that infers ancestry) as a group of birds and all saurischians (dinosaurs) (Weishampel et al 2004).
It is necessary to say that the traditional meaning is intended when the words theropod, feather, bird, and Aves are used in this paper and not the modern meaning influenced by evolutionary ideas.
Archaeopteryx—A Dinosaur with Feathers
The presence of feathers has been the key to classifying an animal as a bird based on the classical, conventional, and traditional taxonomy developed by Linnaeus in the eighteenth century (Gauthier and de Queiroz 2001).
However, various workers have changed and adapted the classification system before and after Darwin (Amorim 2002). The Linnaean classification was based on similarities in creatures, but in contrast, Darwin’s classification is based on their supposed ancestry (Amorim 2002). One can see that the data never changed because the animals and their features have been the same. Instead, the lenses used to interpret them changed.
Some secular and creation scientists have considered Archaeopteryx a reptile or a theropod dinosaur, not a bird (McLain, Petrone, and Speights 2018). The later base this classification on statistical baraminological analyses, which are based on cladistics.
In cladistics, it is hypothesized that the ancestor of birds is in the same ancestral line to which dinosaurs belong. For that reason, it is acceptable to identify a bird as a reptile. Under this new definition, it became correct to state that birds are living dinosaurs. The words “bird” and “reptile” are not meaningful in this method.
However, there are many problems with the bird and dinosaur relationship hypothesis. Two of them will be discussed briefly here.
1. Cladistics is a Method Based on an Evolutionary Worldview
About 100 years after Darwin presented his theory of evolution, the theory of phylogenetic systematics, with its method that has been used in cladistics, was formalized by Willi Hennig (Hennig 1950). Phylogenetic systematics is practiced in the light of evolution, aiming to demonstrate evolutionary history and relationships (Hennig 1965; 1966). And it has become the preferred method for phylogenetic analysis.
In the evolutionary worldview, the main characteristics used for classification are those that allow a path of evolution of certain groups to be revealed. Therefore, the observed features that end up being used in phylogenetic analyses are those that provide some adaptations in the creatures and changes due to new adversities. For taxonomy, this is an important point because it implies that a (supposed) “adversity” (in quotes from the original) can be understood as introducing a new function and purpose.1
Since evolution is the basis on which cladistics was built, a crucial issue must be highlighted. It is based on a sentence written by Ernst Mayr, one of the most influential evolutionist biologists who devoted his studies to evolution and genetics:
Evolution is a historical process that cannot be proven by the same arguments and methods by which purely physical or functional phenomena can be documented. Evolution as a whole, and the explanation of particular evolutionary events must be inferred from observations. Such inferences subsequently must be tested again and again . . . .
However, most inferences made by evolutionists have by now been tested successfully so often that they are accepted as certainties. (Mayr 2001, 13)
Note that he said, “evolution is a historical process.” That means it cannot be observed, tested, or repeated because it happened in the unobserved past. Because of that, as he says, those explanations “must be inferred.” He was also right when he said that the inferences “must be tested.”
That quote also reveals that evolutionist scientists assume that everything came into existence through evolution, so then they develop methods, in this case, cladistics, which is also based on evolution, to test how everything came into existence. In a logical analysis, the evolutionary inferences are tested by evolutionary-based methods and interpreted through an evolutionary worldview to make evolutionary conclusions accepted as certainties. That is circular reasoning. Therefore, this chain of reasoning is faulty, and its conclusions are not logical. In summary, they use evolutionary assumptions to make evolutionary observations tested by evolutionary methods to prove that evolution is true.
So, the first problem with cladistics is that the method of reasoning (used by evolutionary scientists) is faulty. That is, the use of cladistics on Archaeopteryx (as well as on any other fossil set) has methodological problems because it has evolutionary assumptions and faulty logic. The assumptions and the reasoning cannot be stripped out of the method since those assumptions, and the faulty logic are often unrecognized and unknown by the evolutionists. From a creationist perspective, the premises of the evolutionary approach are not biblical. The Scriptures do not teach evolution, and no evolutionary idea should be fitted into the Bible. Even if claimed Christians, creationists, or anti-evolutionists believe otherwise, mere belief does not make it right. Using an authority’s assessment to try to prove a point is also faulty reasoning—an informal logical fallacy called appeal to authority.
This leads to the second problem with cladistics (subjectiveness and arbitrariness), which the history of Archaeopteryx also showcases. The same methods have led various groups to make opposite conclusions. Some conclude that Archaeopteryx was a dinosaur (theropod-sensu stricto) with feathers. Other scientists have placed it more on the bird side of the equation.
Archaeopteryx’s placement is still in dispute in the secular camp. The discovery of Xiaotingia zheng in 2011 helped to shift Archaeopteryx from Avialae to Deinonychosauria (a group of predatory dinosaurs based on some scientists’ view). That result challenged the meaning of Archaeopteryx in light of the supposed transition to birds, and if confirmed, its avialan ancestral condition would need to be reevaluated (Xu et al. 2011). In the following year, Xu and Pol (2013), using the same data but with a different method (a probability-based method), reclassified Archaeopteryx back to Avialae. However, in July 2019, Hartman et al. (2019) placed Archaeopteryx in Deinonychosauria (see fig. 1), whereas, in February 2020, Cau (2020) shifted it back again to Avialae (see fig. 2).
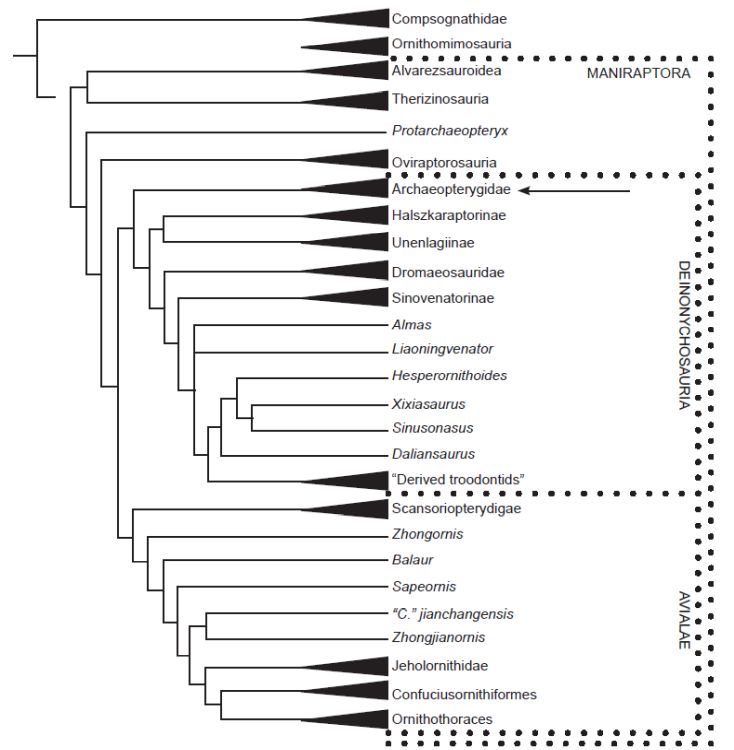
Fig. 1. Phylogenetic analysis diagram by Hartman et al. (2019) placed Archaeopteryx outside their bird group (clade). Arrow indicates the position of Archaeopteryx.
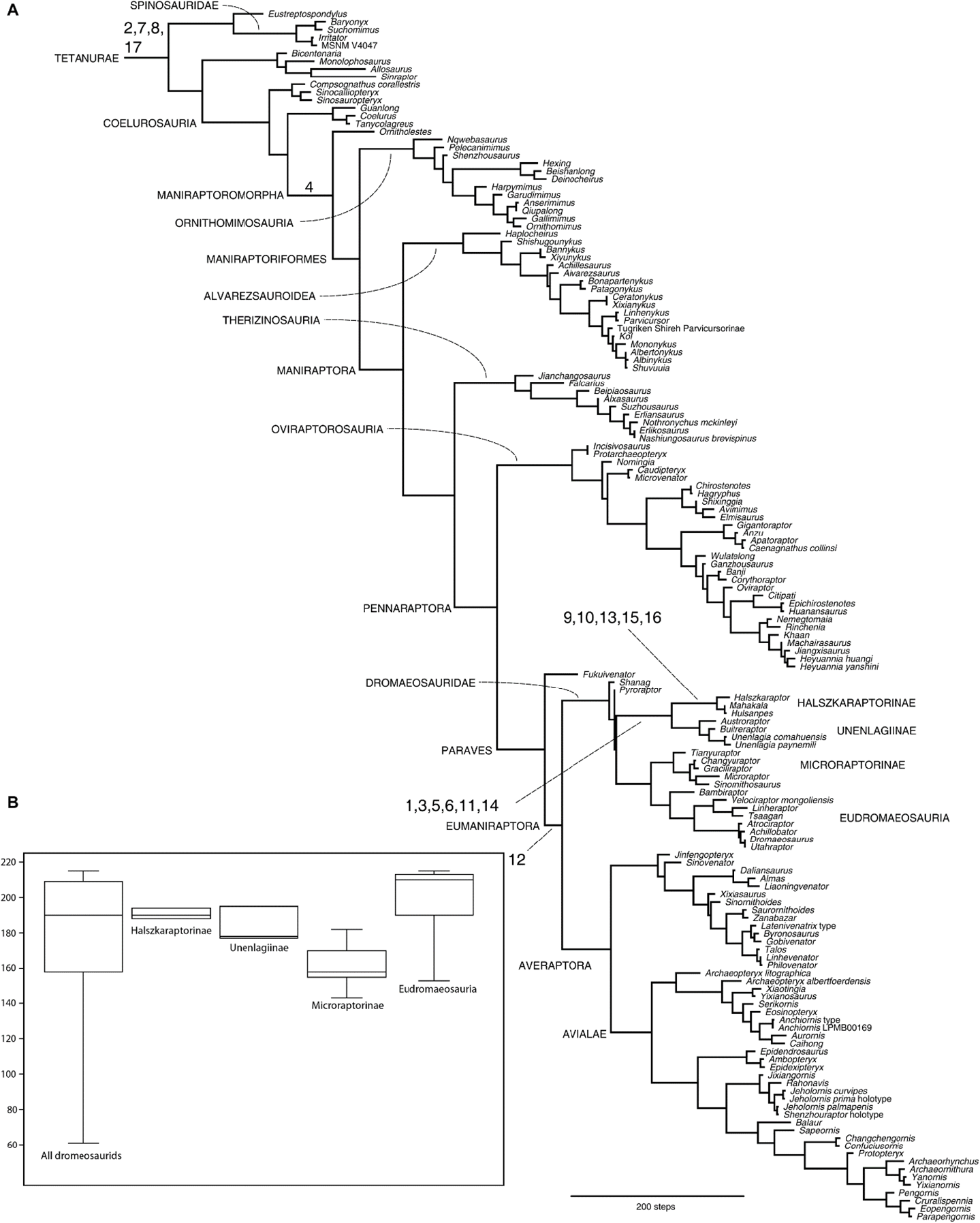
Fig. 2. Phylogeny of maniraptoriforms by Cau (2020) places Archaeopteryx within their bird group (clade). Arrow indicates the position of Archaeopteryx.
That is one example of how subjectively and arbitrarily those hypotheses of evolutionary relationship can be applied. The same data have been interpreted by different groups of scientists with different assumptions, leading not only to distinct conclusions but to opposite conclusions. Consequently, that brings different and opposite implications. Cladistics can place Archaeopteryx in either category—bird or dinosaur. Some scientists blame evolution and conclude that this confusion and lack of resolution are related to rapid rates of evolution in some groups (Brusatte et al. 2014) and the lack of other fossil finds from outside the Cretaceous of China (Hartman et al. 2019).
Some creation scientists have also used the assumptions of cladistics and its results to run baraminological analyses. After an analysis done by creation science opponent Senter in 2010 to prove evolutionary principles through baraminology, some creationist scientists reanalyzed his data and came up with different results from one anothers’ (Cavanaugh 2011; Garner, Wood, and Ross 2013; McLain, Petrone, and Speights 2018).
Cavanaugh (2011) concluded that Archaeopteryx and all theropods (he interpreted Archaeopteryx as a theropod/dinosaur) might be part of the same created kind. In that case, he understood Archaeopteryx as the ancestor of other theropods, not their descendant.
Garner, Wood, and Ross (2013) analyzed some datasets and, in many of their analyses, Archaeopteryx was more correlated with Dromaeosauridae (a family of dinosaurs in some scientists’ view). In one study, it appeared to group with avialans (bird group). They concluded that Archaeopteryx might have been a dromaeosaurid.
McLain, Petrone, and Speights (2018) understood that Archaeopteryx has a high probability of being a part of the deinonychosaur holobaramin, and this case could be resolved with further analysis. They stated in their paper, “Archaeopteryx is so similar to dromaeosaurids and some avialans that multiple creationist studies cannot determine whether it is continuous with traditional dinosaurs or birds.”
For these baraminological analyses, these creationists used statistical methods such as BDC and BDIST. They have devised methods that use statistics to quantify degrees of similarity between organisms’ features and to assign them on these bases to various created kinds. Some problems with this approach will be briefly discussed.
The challenge with this approach is related to the data used in some baraminological analyses. The available published data are taken from the evolutionist literature and can be insufficient, lacking, misinterpreted, misidentified, or misrepresented. It is necessary to understand that the fossil record has its own challenges, and the fossil material is also interpreted. Furthermore, the data used in the method can be arbitrary and subjectively chosen, so the evolutionary bias in the data needs to be considered. If there are problems with the data, that will affect the method’s outcome since it is statistical. Those problems will be reflected in the results, conclusions, and implications. That is, a lack of reliable data and methodological application means a lack of reliable results.
Almost all the studies published using the BDC and BDIST methods have used selected (choosing to show more similarities than differences) and coded (subjectively assigning values to the data) datasets, collected and interpreted by evolutionist scientists based on their evolutionary worldview of cladistics (Sanders and Cserhati 2022).
Evolutionary assumptions cannot be cleansed from their datasets because those datasets are interpreted by evolutionists, who are biased. So, trusting the evolutionist scientists’ results and not considering they are biased is a problem and a challenge for baraminological analyses that use secular datasets. Even though they believe secular scientists are diligent in their research and are presenting factual data, that does not mean they always do so. Critical analyses of all datasets should be done before their use since the data need to be factual to ensure reliable results. This involves checking the bias, which assumptions were applied in the hypothesis of the supposed ancestry history reconstruction, which characters are being analyzed, are the characters correctly identified and interpreted, and ensuring the highest character relevance cutoff number is used.
The cladistics methods are based on subjective choices based on hypotheses. That makes them arbitrary and subjective, so the level of certainty is not very reliable. Since datasets are measured indirectly and based on inferences, they involve evolutionary assumptions and bias (Stadler 2016).
Cladistics has faulty and unbiblical premises that do not fit within a young-earth creation perspective as presented in this paper. So, since the assumptions of cladistics (evolution and common ancestry) are not rooted in the Scriptures, it does not seem reasonable to borrow this method to explain any fossil data within the young-earth creationist framework. If the premises of the method are wrong, it is not logical to assume the conclusiveness of the technique.
In a recent paper, Sanders and Cserhati (2022) list several problems in the methods used in baraminology analyses and baraminological studies. Sanders and Cserhati (2022) also point out that even though those creation scientists who do baraminological studies disagree that a critical analysis of the data is not done, Sanders and Cserhati (2022) demonstrate that one of the tools (relevance statistics) used for that “does not determine whether the character states in the dataset were measured correctly, or in an unbiased fashion. It merely determines what percentage of the characters are present in all taxa contained in the dataset.” The same authors state that the “BDIST also fails an internal critique” and conclude that a critical reevaluation of the method needs to be addressed.
2. Cladistics Uses Different and Ambiguous Terms and Definitions
Cladistics terms and definitions are used in a very subjective and arbitrary way. A quick search of the definition of “bird” will lead you to find that birds are feathered theropod dinosaurs and are thus living dinosaurs. Birds are reptiles in cladistics.
As one can see, the word “bird” today does not mean what we used to know and understand about birds. Because of evolutionary assumptions, the definitions of the words “bird,” “aves,” and even “feathers” have changed to fit into an evolutionary perspective. Those words have been redefined through an evolutionary lens. That is, anywhere they point or see, evolution is the reason and the explanation for everything.
One example of this is the term “Avialae,” which was defined and named by Gauthier (1986), who redefined it in 2001 (Gauthier and de Queiroz 2001). Avialae, depending on the definition used for it (whether based on characters or based on ancestry), can “split” or “lump,” moving apart groups of creatures or gathering them together, respectively.
Another redefined term is “Aves,” despite its etymological meaning. Avis in Latin means birds, but because of phylogenetic systematics, that is not its meaning anymore. Aves and birds are not synonymous (Gauthier 1986). Because of cladistics, five different groups have been named Aves in a formal manner and six in an informal way. Gauthier and de Queiroz (2001) demonstrated the problem with five to six different definitions for the same term.
They attempted to solve the problem by developing more different definitions. In their view, only four of those groups should be named Aves, but that did not bring any resolution. Then, they decided on what the definition of those groups would be based, that is, how they should be differentiated and named.
Because of cladistics, this question cannot be easily answered regarding the issue of what a bird is. It will depend on:
- (1) If the definition emphasizes composition over character;
- (2) If the definition is based on composition, then whether it is stem-based or node-based?
- (3) If the definition is based on character, then which character or characters are more or the most important?
- (4) If the most important characters can be derived (shared), if not, which one should be; and
- (5) Which author’s or research group’s conclusion will be followed as a convention.
That is, depending on any one of the options listed above, cladistics can produce different classifications. Cladistics has subjective applications because it is a hypothesized model, which means that different research groups have different hypotheses on this topic and build different models of explanations of the supposed evolutionary relationships between organisms through inferences that lead to different, and in many cases, opposite conclusions.
Because of definitions and terms being changed, birds have been included within the Dinosauria group (Benton 2004). So, one can say that birds are theropods or use the expression living/flying theropod or living/flying dinosaurs for birds. The terms have become so plastic as to lose connection with the realities of animal anatomy. In this way, these terms replace usefulness in classification for usefulness in propagating evolution.
The following is a sequence of groups for didactic purposes, yet it is not a representation of cladistics:
Theropod-Coelurosauria-Maniraptora-Pennaraptora-Paraves-(Eumaniraptora is also used sometimes. Despite being considered synonymous with Paraves, some phylogenetic analyses demonstrate they might group differently)-Avialae-Aves.
The main point here is that those definitions are arbitrary and ever-changing. They will depend on which phylogenetic hypotheses were made, what they were based on, and the definition used. Another example of that is related to the word “feather.” If the feather is defined as filaments, then they appear in basal coelurosaurs such as Sinosauropteryx. If a feather is defined as pennaceous, then it appears in maniraptorans. The definition of feather changes everything, whether more like Prum (1999), who proposed and hypothesized that a feather evolved through a series of stages, or more like the classical and traditional way we all understand what a feather is and has been found in the fossil record, complete and functional.
So, for definition purposes, it is essential to highlight that the word “bird” used here is defined in the classical, traditional, Linnean way. That is, having actual modern-looking feathers. It is also necessary to define feathers. (Yes, because of an evolutionary worldview, simple things like a bird and feathers must be clearly stated now.) And feather means a complete and functional structure with no evolutionary stages and that only birds are known to possess.
Added to the criticism of the applications of evolutionary assumptions and the circular reasoning process used in testing their inferences, there are even more criticisms of phylogenetic systematics that cannot be discussed here. Phylogenetic systematics is a method that not all researchers accept because of the subjectiveness and inconsistencies in its concepts and applications, data challenges, and the limited understanding of the organisms’ features needed for analysis. These problems indeed produce confusing and unstable conclusions resulting from that method.
Archaeopteryx—A New Attempt
Dr. Marc Surtees has proposed a new approach to Archaeopteryx. His article, published in 2021, suggested a new definition for the term bird (Surtees 2021).
The reason for redefining the term bird is because Dr. Surtees seems to see that the reports of feathers on other theropods are convincing. He based that on an article written by McLain, Petrone, and Speights (2018). Dr Surtees also mentioned that Archaeopteryx shared features with what he called the “bird-like” theropod. He listed some of the same features that Wellnhofer (2009) presented, which I am discussing in this paper.
He also mentioned another feature that I will discuss here: the presence of air sacs and a flow-through lung in a sauropod species, Majungasaurus atopus.
The presence of air-filled cavities (pneumaticity) in the post-cranial vertebrae of many dinosaurs has been known since around 1870. Pneumaticity in the postcranial area is also described in pterosaurs. This feature has been considered functionally related to a specialization to reduce weight (Benson et al. 2012; O’Connor 2004; 2009).
Robert T. Bakker was mentored by John Ostrom, the one who revived the idea of the relationship between birds and dinosaurs after studying Deinonychus. Bakker’s articles gave support to the notion that dinosaurs were warm-blooded. In 1972, Bakker proposed (emphasis mine) that the cavities in the vertebrae called postcranial skeletal pneumaticity (PSP) found in dinosaurs were for air sacs. He said dinosaurs “probably had an avian-type lung with unidirectional flow” (Bakker 1972).
Nonetheless, Schachner, Hutchinson, and Farmer (2013) say in their article on Nile crocodiles published in 2013:
Many other studies have also tried to use pneumaticity to sort out respiratory anatomy and the presence or absence of specific patterns of flow in the lungs of extinct vertebrates (e.g., O’Connor and Claessens, 2005; O’Connor, 2006; O’Connor, 2009; Wedel, 2006; Wedel, 2007; Wedel, 2009). Yet postcranial pneumaticity has been purported to be equivocal evidence at best for patterns of air flow, lung efficiency, thermoregulatory strategies, and exercise capacities because pneumaticity has no known function in respiration or gas exchange (Farmer, 2006). Here we have shown that Nile crocodiles neither have postcranial pneumaticity nor air sacs and yet have lungs with truly flow-through ventilation. Hence unidirectional ventilatory flow (a flow-through lung in physiological terms) is possible in an ectothermic animal without pneumaticity and without air sacs. This emphasizes that bronchial anatomy, air sac anatomy, and ventilatory patterns can be decoupled from each other in archosaurs and should not be presumed to be correlated in simple ways.
Majungasaurus vertebrae have features that imply (emphasis mine) the presence of air sacs because of cavities in them. This assumption relies on the evolutionary-based interpretation proposed by Bakker. Also, Butler, Barret, and Gower (2012), in their research on a reassessment of the evidence for PSP, say that the evidence for postcranial skeletal pneumaticity can be vascular in origin. That is, some of the evidence for PSP can be misidentified and misinterpreted.
So, based on what has been presented about Bakker’s evolutionary-based proposal in 1972, Schachner, Hutchinson, and Farmer (2013), and Butler, Barrett, and Gower (2012), we can better understand the background of the pneumaticity and air sacs related topics regarding their presence in dinosaurs. It is necessary to consider all that and discern that the consequence of this idea aims to support the theory of the relationship between birds and dinosaurs.
Dr. Surtees uses evolutionary definitions and relies on baraminological analyses (which are also based on, and influenced by, evolutionary bias and have methodological problems), justifying the need for a change in the term bird. That alone does not make his attempt incorrect but unnecessary, since none of the reasons he listed for this change is reasonable. Yet, his work and ideas have influenced students to keep propagating and basing their views on evolutionary-based definitions. That is the case for an article published by a geology student in the New Creation Blog (Ryan 2022).
Even if we granted the presence of PSP or soft tissue of air sacs in dinosaurs, that would not make it necessary to have the term bird redefined. God is the Creator and Designer of all creatures, so we expect to find common design features in different groups of animals.
Archaeopteryx—A Bird
Archaeopteryx was first described and classified as a bird by Richard Owen in 1863. Distinctive bird features have supported this classification in the 12 body specimens found of Archaeopteryx.
All Archaeopteryx specimens are associated with feathers except for one, the twelfth specimen. The presence of feathers is not the only characteristic but one of the main ones because feathers are central to the definition of aves since only birds are known for possessing feathers (Brush 1996, 2001; Lee and Spencer 1997; Paul 1988). The structure and arrangement of Archaeopteryx’s feathers look just like the ones found in modern birds—complete, complex, and showing full functionality for flight. Also, the morphology of the cranium, inner ear, sclerotic plates, and ratio of lower to the upper leg are some of the features that uniquely belong to birds that are also present in Archaeopteryx (Wellnhofer 2009).
Archaeopteryx had avian wings arranged with primary and secondary asymmetric feathers (Feduccia 2020). Their macro and microstructures are like the feathers in birds we see today. These properties provide information enabling us to infer that Archaeopteryx was likely a glider with flap capability over short distances. A study by Voeten et al. (2018) showed the wing bone geometry of some specimens of Archaeopteryx. He stated that Archaeopteryx “exhibits a combination of cross-sectional geometric properties uniquely shared with volant birds.” Even though a paper by Foth, Tischlinger, and Rauhut (2014) presented the idea that the symmetric feathers found on the eleventh specimen were likely just for display, that does not compete with the idea they could also have been used for flight. Some birds have the same symmetric feathers and can glide and flap (for example, the kakapo Strigops hadroptilus) (Feduccia 2020). Many other studies on the structure of Archaeopteryx’s brain (Alonso et al. 2004) and its skeletal adaptations (Feduccia 2020) reveal that Archaeopteryx was capable of flying. One of those studies (Kundrát et al. 2019) states, “However, the overall morphology of Archaeopteryx lithographica, as demonstrated by all of the Bavarian specimens, clearly indicates a capacity for aerial travel (whether gliding or flapping) . . . .” Added to that, the range of motion of the forelimb is the same as that in modern birds (fig. 3). The same pattern of wing folding was also present in Archaeopteryx.
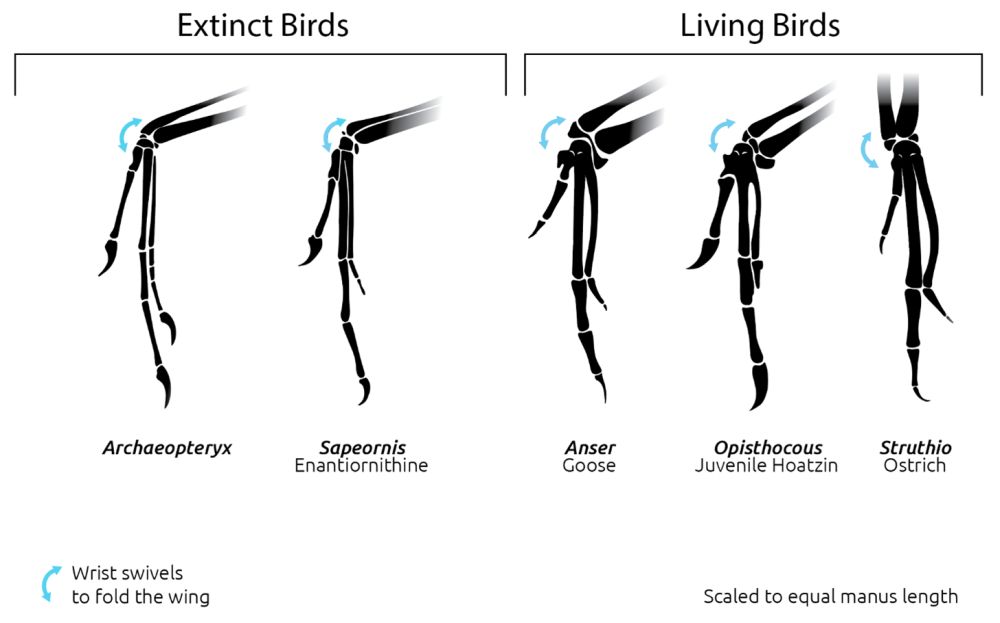
Fig. 3. Forelimb motion range comparison diagram created by Joel Leineweber using the software Blender 3.0 (2021) and Procreate 5.2 (2022).
Some reconstructions of Archaeopteryx show its separated fingers in a more predatory position (fig. 4), but later researchers discovered the outer and middle fingers were united instead (Feduccia 2020). Hartman (2013) in Hendrickx, Hartman, and Mateus 2015 presented Archaeopteryx in a more theropod pose. Even though he has not changed much in his new version (2022) of Archaeopteryx, he has the arm position more folded as a wing, and the body is a little bit upright (fig. 5).
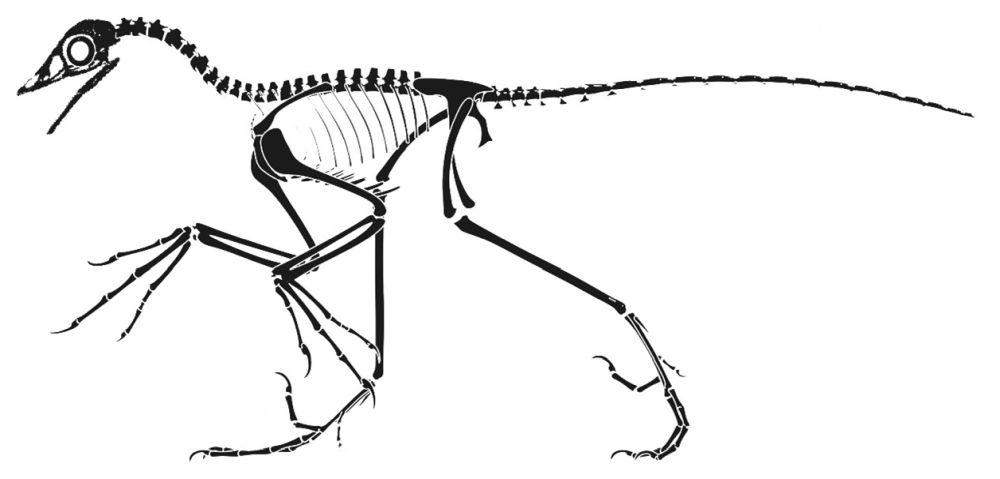
Fig. 4. Skeletal drawings of Archaeopteryx on a dinosaur pose adapted from Hartman (2013) in Hendrickx, Hartman and Mateus (2015) by Joel Leineweber using the software Blender 3.0 (2021) and Procreate 5.2 (2022).
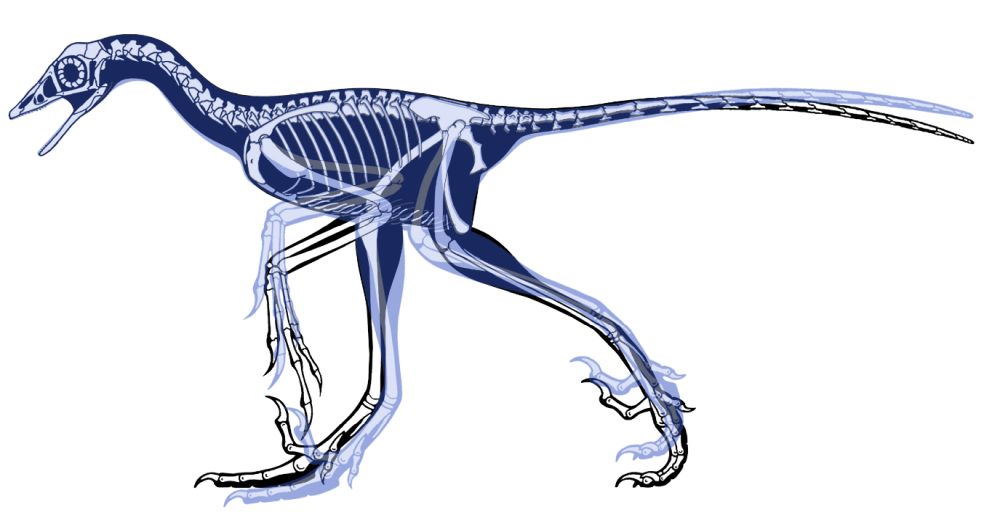
Fig. 5. Skeletal drawings comparison of Archaeopteryx on a dinosaur pose from Hartman’s version 2013 and 2022 adapted by Joel Leineweber using Adobe Photoshop 23 (2021). In black is Hartman’s 2013 version of Archaeopteryx, and in blue is Hartman’s 2022 version of Archaeopteryx.
The number of caudal vertebrae in Archaeopteryx also shows a difference between birds and theropods. Archaeopteryx has around 21–23 vertebrae, while theropod dinosaurs have 30–40 (fig. 6) (Senter et al. 2012; Wellnhofer 2009).
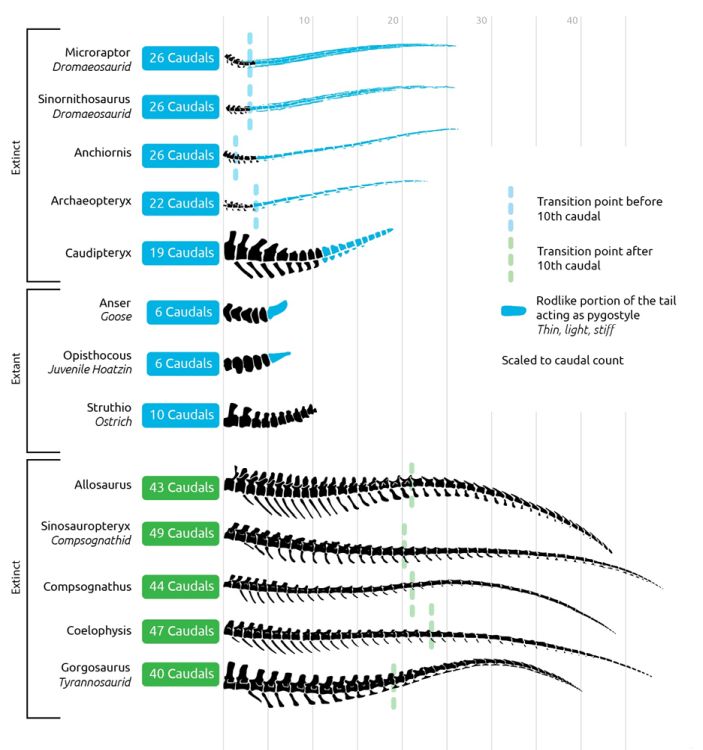
Fig. 6. Tail comparison diagram (credit: Joel Leineweber). In blue, supposed dinosaurs and living birds show the same tail characteristics in clear contrast to those of dinosaurs, shown in green.
Archaeopteryx has been considered a carnivore like a dinosaur because of the presence of teeth and having no beak (Wellnhofer 2009). However, this does not seem to be the case for Archaeopteryx. Since nothing has been preserved in the stomach of the specimens of Archaeopteryx, it is necessary to check the indirect evidence to infer any information on its diet. Archaeopteryx’s teeth do not show features required for cutting flesh (Wellnhofer 2009). So, as some evolutionists want to pose it, it is reasonable to ponder the possibility of Archaeopteryx not being a predatory carnivorous animal.
Seeing in the specimens of Archaeopteryx the presence of those features that are diagnostic of birds, as well as considering all the problems with the methodological system of classification with its assumptions and biased evolutionary interpretations, understanding Archaeopteryx as a bird becomes straightforward (fig. 7).
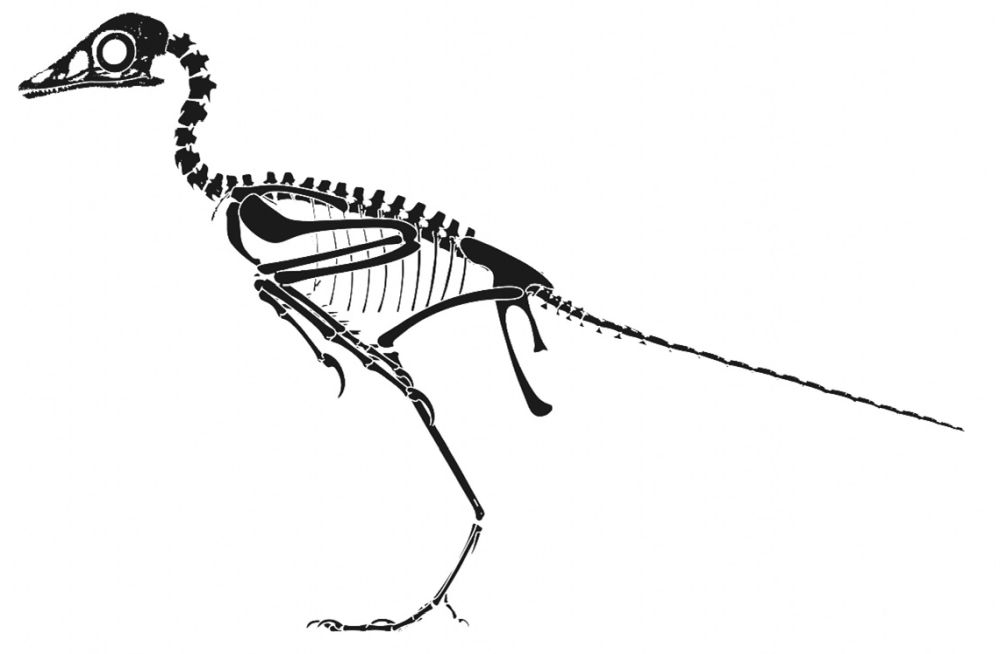
Fig. 7. Skeletal drawings of Archaeopteryx on a bird pose adapted from Wellnhofer (2009) by Joel Leineweber using the software Blender 3.0 (2021) and Procreate 5.2 (2022).
Conclusions
Creation and secular scientists remain divided on Archaeopteryx’s classification. As this paper demonstrates, the observable data is the same, but the scientists that interpret them have different starting assumptions. Young-earth creationists believe God created birds and dinosaurs; evolutionists believe that birds and dinosaurs share an ancestry history. That is why the interpretations of the data sometimes arrive at opposite conclusions.
The attempts to reclassify, redefine, and reinterpret Archaeopteryx will not make it become a dinosaur. A bird will never be a dinosaur, regardless of what, how, or who states it. Birds have distinctive features like feathers, arm bone anatomy, and pygostyle that are irreconcilable to those of dinosaurs. Just like the modern bird hoatzin, Archaeopteryx was created and designed to have features that did precisely what Archaeopteryx needed. Some of them we might not understand why or how, but our lack of understanding should not make us surpass biblical limits. God’s Word is clear and definitive—birds were created on Day Five and dinosaurs among the land animals on Day Six of the Creation Week. God created birds before He created dinosaurs, and not the contrary. They are different kinds created on different days of the Creation Week. Thus, calling a bird a dinosaur presents both scientific and biblical problems.
In sum, the classification system that evolutionists use is characterized by:
- (1) A faulty chain of reasoning,
- (2) Evolutionary assumptions, and
- (3) Methodological problems such as:
- (a) Subjective, arbitrary and confusing definitions,
- (b) Arbitrary and confusing application of the definitions,
- (c) Data challenges,
- (d) Subjective and arbitrary interpretation of data, and
- (e) Subjective and arbitrary application of data.
All these considerations lead to the conclusion that there is no reasonable basis or explanation for agreeing with evolutionary assumptions that claim Archaeopteryx to be a missing link, neither with the cladistic approach for its placement nor with the baraminological approach that follows the premises of cladistic taxonomy and presents challenges in the application of its methodology.
In summary, there is no reasonable explanation to conclude that Archaeopteryx:
- (1) is a transitional form, or
- (2) is related to dinosaurs, or
- (3) is a dinosaur with feathers.
Birds are considered dinosaurs due to reliance on the assumptions of the evolutionary worldview and its method of classification, cladistics.
So, “Is Archaeopteryx then actually a feathered dinosaur? This remains a question of definition” (Wellnhofer 2009, 166). That is the cladistics approach with Archaeopteryx. In contrast, in a biblical worldview, as well as on logical grounds, the anatomical features of the skeleton and skull, the presence of feathers (which is a key diagnostic for bird identification), and following the classical, traditional Linnean classification and reasoning, it is concluded that there is no reason for Archaeopteryx to be anything other than a bird.
References
Alonso, Patricio Domínguez, Angela C. Milner, Richard A. Ketcham, M. John Cookson, and Timothy B. Rowe. 2004. “The Avian Nature of the Brain and Inner Ear of Archaeopteryx.” Nature 430, no. 7000 (5 August): 666–669.
Amorim, Dalton de Souza. 2002. Fundamentos de Sistemática Filogenética, Ribeirão Preto: Holos.
Bakker, Robert. T. 1972. “Anatomical and Ecological Evidence of Endothermy in Dinosaurs.” Nature 238, no. 5359 (14 July): 81–85.
Benson, Roger B. J., Richard J. Butler, Matthew T. Carrano, and Patrick M. O’Connor. 2012. “Air-Filled Postcranial Bones in Theropod Dinosaurs: Physiological Implications and the ’Reptile’-Bird Transition.” Biological Reviews 87, no. 1 (February): 168–193.
Benton, Michael J. 2004. “Origin and Relationships of Dinosauria.” In The Dinosauria, edited by David B. Weishampel, Peter Dodson, and Halszka Osmólska, 7–19. Berkeley, California: University of California Press.
Brusatte, Stephen L, Graeme T. Lloyd, Steve C. Wang, and Mark A. Norell. 2014. “Gradual Assembly of Avian Body Plan Culminated in Rapid Rates of Evolution Across the Dinosaur-Bird Transition.” Current Biology 24, no. 20 (20 October): 2386–2392.
Brush, A. H. 1996. “On the Origin of Feathers.” Journal of Evolutionary Biology 9, no. 2 (March): 131–140.
Brush, A. H. 2001. “The Beginnings of Feathers.” In New Perspectives on the Origin and Early Evolution of Birds: Proceedings of the International Symposium in Honor of John H. Ostrom, edited by Jacques Gauthier and Lawrence F. Gall, 171–179. New Haven, Connecticut: Peabody Museum of Natural History, Yale University.
Butler Richard J., Paul M. Barrett, and David J. Gower. 2012. “Reassessment of the Evidence for Postcranial Skeletal Pneumaticity in Triassic Archosaurs, and the Early Evolution of the Avian Respiratory System.” PLoS ONE 7, no. 3 (March 28): e34094. https://doi.org/10.1371/journal.pone.0034094.
Carney, Ryan M., Helmut Tischlinger, and Matthew D. Shawkey. 2020. “Evidence Corroborates Identity of Isolated Fossil Feather as a Wing Covert of Archaeopteryx.” Science Reports 10: 15593. https://doi.org/10.1038/s41598-020-65336-y.
Cau, Andrea. 2020. “The Body Plan of Halszkaraptor escuilliei (Dinosauria, Theropoda) is not a Transitional Form Along the Evolution of Dromaeosaurid Hypercarnivory.” PeerJ 8 (February 25): e8672. https://doi.org/10.7717/peerj.8672.
Cavanaugh, David. 2011. “An ANOPA Study of Coelurosaurian Theropods.” Journal of Creation Theology and Science Series B: Life Science 1, no. 2 (27 July): 18.
Claessens, Leon. 2004. “Dinosaur Gastralia: Origin, Morphology, and Function.” Journal of Vertebrate Paleontology 24, no. 1 (March): 89–106.
Dames, Wilhelm. 1884. “Üeber. Archaeopteryx.” Palæontologische Abhandlungen 2:119–196. Berlin, Germany: G. Reimer.
Dames, Wilhelm. 1897. “Über Brustbein, Schulter-und Beckengürtel der Archaeopteryx.” Sitzungsberichte der Königlichpreussischen Akademie der Wissenschaften zu Berlin 38: 818–834.
Dames, R. 1927. “Werner von Siemens und der Archaeopteryx.” Nachrichten des Vereins der Siemens-Beamten Berlin e. V.: 233–234.
Elżanowski, Andrzej. 2001. “A New Genus and Species for the Largest Specimen of Archaeopteryx.” Acta Palaeontologica Polonica 46, no. 4 (October) :519–532.
Elżanowski, Andrzej. 2002. “Archaeopterygidae (Upper Jurassic of Germany).” In Mesozoic Birds. Above the Heads of Dinosaurs, edited by Luis M. Chiappe, and Lawrence M. Witmer, 129–159. Berkeley, California: University of California Press.
Elżanowski, Andrzej, and Peter Wellnhofer. 1996. “Cranial Morphology of Archaeopteryx: Evidence from the Seventh Skeleton.” Journal of Vertebrate Paleontology 16, no. 1 (March 19): 81–94.
Feduccia, Alan. 2020. Romancing the Birds and Dinosaurs: Forays in Postmodern Paleontology. Irvine, California: Brown Walker Press.
Foth, Christian, and Oliver W. M. Rauhut. 2017. “Re-evaluation of the Haarlem Archaeopteryx and the Radiation of Maniraptoran Theropod Dinosaurs.” BMC Evolutionary Biology 17, no. 1 (December 2): 236–252.
Foth, Christian, Helmut Tischlinger, and Oliver W. M. Rauhut. 2014. “New Specimen of Archaeopteryx Provides Insights into the Evolution of Pennaceous Feathers.” Nature 511, no. 7507 (3 July): 79–82.
Frickhinger, K. A. 1999. Die Fossilien von Solnhofen. Vol. 2. The Fossils of Sonhofen. Korb, Germany: Goldschneck.
Garner, Paul, Todd C. Wood, and Marcus Ross. 2013. “Baraminological Analysis of Jurassic and Cretaceous Avialae.” In Proceedings of the Seventh International Conference on Creationism, edited by Mark Horstemeyer, article 7. Pittsburgh, Pennsylvania: Creation Science Fellowship.
Gauthier, Jacques. 1986. “Saurischian Monophyly and the Origin of Birds.” Memoirs of the California Academy of Sciences 8: 1–55.
Gauthier, Jacques, and Kevin de Queiroz. 2001. “Feathered Dinosaurs, Flying Dinosaurs, Crown Dinosaurs, and the Name Aves.” In New Perspectives on the Origin and Early Evolution of Birds: Proceedings of the International Symposium in Honor of John H. Ostrom, edited by Jacques Gauthier, and Lawrence F. Gall, 7–41. New Haven, Connecticut: Peabody Museum of Natural History, Yale University.
Grajal, Alejandro. 1995. “Structure and Function of the Digestive Tract of the Hoatzin (Opisthocomus Hoazin): A Folivorous Bird with Foregut Fermentation.” The Auk 112, no. 1 (1 January): 20–28.
Griffiths, P. J. 1996. “The Isolated Archaeopteryx Feather.” Archaeopteryx 14: 1–26.
Hartman Scott, Mickey Mortimer, William R. Wahl, Dean R. Lomax, Jessica Lippincott, and David M. Lovelace. 2019. “A New Paravian Dinosaur from the Late Jurassic of North America Supports a Late Acquisition of Avian Flight.” PeerJ 7 July 10): e7247. https://doi.org/10.7717/peerj.7247.
Hartman Scott. 2022. “Dr. Scott Hartman’s Skeltal Drawing.com.” https://www.skeletaldrawing.com/theropods/archaeopteryx.
Heilmann, Gerhard. 1926. The Origin of Birds. London, United Kingdom: Witherby.
Hendrickx, Christophe, Scott A. Hartman, and Octávio Mateus. 2015. “An Overview of Non-Avian Theropod Discoveries and Classification.” PalArch’s Journal of Vertebrate Palaeontology 12, no. 1: 1–73.
Hennig, Willi. 1950. Grundzüge einer Theorie der Phylogenetischen Systematik. Berlin, Germany: Deutscher zentralverlag.
Hennig, Willi. 1965. “Phylogenetic Systematics.” Annual Review of Entomology 10 (January): 97–116.
Hennig, Willi. 1966. Phylogenetic Systematics. Champaign, Illinois: University of Illinois Press.
Howgate M. E. 1984. “The Teeth of Archaeopteryx and a Reinterpretation of the Eichstätt Specimen.” Zoological Journal of the Linnean Society 82, nos. 1-2 (September): 159–175.
Howgate, Michael E. 1985. “Problems of the Osteology of Archaeopteryx. Is the Eichstätt specimen a distinct genus?” In The Beginning of Birds. Proceedings of the International Archaeopteryx Conference Eichstätt, edited by Max K. Hecht, John H. Ostrom, Gunter Viohl, and Peter Wellnhofer, 105-112. Eichstätt, Germany: Freunde des Jura Museums.
Huxley, Thomas Henry. 1868. “Remarks upon Archaeopteryx lithographica.” Proceedings of the Royal Society of London 16 (31 December): 243–248.
Huxley, Thomas H. 1868. “On the Animals Which are Most Nearly Intermediate Between Birds and Reptiles.” Annals and Magazine of Natural History 4: 66-75.
Kaye, Thomas G., Michael Pittman, Gerald Mayr, Daniela Schwarz, and Xing Xu. 2019. “Detection of Lost Calamus Challenges Identity of Isolated Archaeopteryx Feather.” Scientific Reports 9 (4 February): 1182. https://doi.org/10.1038/s41598-018-37343-7.
Kundrát, Martin, John Nudds, Benjamin P. Kear, Junchang Lü, and Per Ahlberg. 2019. “The First Specimen of Archaeopteryx From the Upper Jurassic Mörnsheim Formation of Germany.” Historical Biology 31, no. 1 (24 October): 3–63.
Lambrecht, Kálmán. 1933. Handbuch der Palaeornithologie. Gebrüder Borntraeger: Berlin.
Lee, Michael Y. S., and Patrick S. Spencer. 1997. “Crown- Clades, Key Characters and Taxonomic Stability: When is an Amniote not an Amniote?” In Amniote Origins: Completing the Transition to Land, edited by Stuart S. Sumida and Karen L. M. Martin, 61–84. San Diego, California: Academic Press.
Mäuser, M. 1997. “Der achte Archaeopteryx.” Fossilien 3: 156– 157.
Mayr, Ernst. 2001. What Evolution Is. New York, New York: Basic Books.
Mayr, Franz X. 1973. “Ein neuer Archaeopteryx-Fund.” Paläontologische Zeitschrift 47, nos. 1–2 (June): 17–24.
Mayr, Gerald. 2005. “Das zehnte Skelettexemplar eines Archaeopterygiden.” Archaeopteryx 23: 1–2.
Mayr, Gerald, Burkhard Pohl, and D. Stefan Peters. 2005. “A Well-Preserved Archaeopteryx Specimen with Theropod Features.” Science 310, no. 5753 (2 December): 1483–1486.
Mayr, Gerald, Burkhard Pohl, Scott Hartman, D. Stefan Peters. 2007. “The Tenth Skeletal Specimen of Archaeopteryx.” Zoological Journal of the Linnean Society 149, no. 1 (January): 97-116.
McLain, Matthew A., Matt Petrone, and Matthew Speights. 2018. “Feathered Dinosaurs Reconsidered: New Insights from Baraminology and Ethnotaxonomy.” In Proceedings of the Eighth International Conference on Creationism, edited by John H. Whitmore, 472–515. Pittsburgh, Pennsylvania: Creation Science Fellowship.
Meyer, Hermann von. 1857. “Beiträge zur näheren Kenntnis fossiler Reptilien.” In Neues Jahrbuch für Mineralogie, Geologie, Geognosie und Petrefakten-Kunde, edited by K. C. von Leonhard and H. G. Bronn, 532–543. Stuttgart, Germany: E. Schweizerbart’sche Verlagshandlung und Druckerei.
Meyer, Hermann von. 1859. Zur Fauna der Vorwelt. Vol. 4. Reptilien aus dem Lithographischen Schiefer des Jura in Deutschland und Frankreich. Germany: Frankfurt a.M.
O’Connor, Jingmai K., and Luis M. Chiappe. 2011. “A Revision of Enantiornithine (Aves: Ornithothoraces) Skull Morphology.” Journal of Systematic Palaeontology 9, no. 1 (6 January): 135–157.
O’Connor, Jingmai K., Thomas A. Stidham, Jerald D. Harris, Matthew C. Lamanna, Alida M. Bailleul, Han Hu Min Wang, and Hai-Lu You. 2022. “Avian Skulls Represent a Diverse Ornithuromorph Fauna from the Lower Cretaceous Xiagou Formation, Gansu Province, China.” Journal of Systematics and Evolution 60. https://doi.org/10.1111/jse.12823.
O’Connor, Patrick M. 2004. “Pulmonary Pneumaticity in the Postcranial Skeleton of Extant Aves: A Case Study Examining Anseriformes.” Journal of Morphology 261, no. 2 (August): 141–161.
O’Connor, Patrick M. 2006. “Postcranial Pneumaticity: An Evaluation of Soft-Tissue Influences on the Postcranial Skeleton and the Reconstruction of Pulmonary Anatomy in Archosaurs.” Journal of Morphology 267, no. 10 (October): 1199–1226.
O’Connor, Patrick Michael. 2009. “Evolution of Archosaurian Body Plans: Skeletal Adaptations of an Air-Sac-Based Breathing Apparatus in Birds and Other Archosaurs.” Journal of Experimental Zoology 311A, no. 8 (1 October) Special Issue: Sauropsid Respiration: 629–646.
O’Connor, Patrick M. and Leon P. A. M. Claessens. 2005. “Basic Avian Pulmonary Design and Flow-Through Ventilation in Non-Avian Theropod Dinosaurs.” Nature 436, no. 7048 (July 14): 253–256.
Ostrom, John H. 1969. “Osteology of Deinonychus antirrhopus, an Unusual Theropod from the Lower Cretaceous of Montana.” Bulletin of the Peabody Museum of Natural History 30:1–165.
Ostrom, John H. 1972. “Description of the Archaeopteryx specimen in the Teyler Museum, Haarlem.” Proceedings Koninklijk Nederlandse Akademie van Wetenschappen B, Physical Sciences 75, no. 4: 289–305.
Ostrom, John H. 1976. “Archaeopteryx and the Origin of Birds.” Biological Journal of the Linnean Society 8, no. 2 (June): 91–182.
Ostrom, John H. 1991. “The Question of the Origin of Birds.” In Origins of the Higher Groups of Tetrapods: Controversy and Consensus, edited by Hans-Peter Schultz, and Linda Trueb, 467–484. Ithaca, New York: Comstock Publishing Associates.
Ostrom, John H. 1994. “On the Origin of Birds and of Avian Flight.” Short Courses in Paleontology. Vol. 7. Major Features of Vertebrate Evolution: 160–177.
Owen, Richard. 1862. “On the Fossil Remains of a Long-Tailed Bird (Archeopteryx macrurus, Ow.) from the Lithographic Slate of Solenhofen.” Proceedings of the Royal Society of London 12: 271–273.
Owen, Richard. 1863. “On the Archaeopteryx of Von Meyer with a Description of the Fossil Remains of a Long-Tailed Species, From the Lithographic Stone of Solenhofen.” Philosophical Transactions of the Royal Society of London 153 (1 January): 33–47.
Parker, William Kitchen. 1891. “On the Morphology of a Reptilian Bird, Opisthocomus cristatus.” Transactions of the Zoological Society of London 13: 43–84.
Paul, Gregory S. 1988. Predatory Dinosaurs of the World: A Complete Illustrated Guide. New York, New York: Simon and Schuster.
Petroniević, Branislav. 1921. Ueber das Becken, den Schultergürtel und einige andere Teile der Londoner Archaeopteryx. Geneva, Switzerland: Georg & Co.
Prum, Richard O. 1999. “Development and Evolutionary Origin of Feathers.” Journal of Experimental Zoology 285, no. 4 : 291–306.
Rauhut, Oliver W. M., Christian Foth, and Helmut Tischlinger. 2018. “The Oldest Archaeopteryx (Theropoda: Avialiae): A New Specimen From the Kimmeridgian/Tithonian Boundary of Schamhaupten, Bavaria.” PeerJ 6 (June 17): e4191. doi.org/10.7717/peerj.419.
Röper, M. 2004. “Kurznotiz: Nachweis von Überresten eines neuen Exemplars des Urvogels Archaeopteryx aus Solnhofen.” Archaeopteryx 22: 1–2.
Ryan, Christian. 2022. “Archaeopteryx: Just a Weird Perching Bird?” New Creation, July 20. https://newcreation.blog/archaeopteryx-just-a-weird-perching-bird/.
Sanders, Harry F. III. 2019. “Hoatzin Bird: Evidence Against Evolutionary Ideas.” Answers in Depth 14. https://answersingenesis.org/birds/hoatzin-bird-evidence-against-evolutionary-ideas/.
Sanders, Harry F. III, and Matthew F. Cserhati. 2022. “Statistics, Baraminology, and Interpretations: A Critical Evaluation of Current Morphology-Based Baraminology Methods.” Creation Research Society Quarterly 58, no. 3 (Winter): 175–192.
Schachner, Emma, John R. Hutchinson, and C. G. Farmer. 2013. “Pulmonary Anatomy in the Nile Crocodile and the Evolution of Unidirectional Airflow in Archosauria.” PeerJ 1 (March 26): e60. https://peerj.com/articles/60/.
Senter, Phil. 2010. “Using Creation Science to Demonstrate Evolution: Application of a Creationist Method for Visualizing Gaps in the Fossil Record to a Phylogenetic Study of Coelurosaurian Dinosaurs.” Journal of Evolutionary Biology 23, no. 8 (August): 1732–1743.
Senter Phil, James I. Kirkland, Donald D. DeBlieux, Scott Madsen, and Natalie Toth. 2012. “New Dromaeosaurids (Dinosauria: Theropoda) from the Lower Cretaceous of Utah, and the Evolution of the Dromaeosaurid Tail.” PLoS One 7, no. 5 (May 15): e36790. https://doi.org/10.1371/journal.pone.0036790.
Stadler, Robert. 2016. The Scientific Approach to Evolution. CreateSpace Independent Publishing Platform.
Stephan, Burkhard. 1987. Urvögel Archaeopterygiformes. 3rd ed. Wittenberg, Germany: Ziemsen.
Surtees, Marc. 2021. “Is It a Bird? A Critical Analysis of Feathered Fossils.” Journal of the Biblical Creation Trust 3: 12–19.
Tischlinger, H. and K.-H. Scharf. 1998. “Das 8. Archaeopteryx- Exemplar.” Praxis der Naturwissenschaften Biologie 47, no. 5: 1–2.
Voeten, Dennis F. A. E., Jorge Cubo, Emmanuel de Margerie, Martin Röper, Vincent Beyrand, Stanislav Bureš, Paul Tafforeau, and Sophie Sanchez. 2018. “Wing Bone Geometry Reveals Active Flight in Archaeopteryx.” Nature Communications 9, no. 1. https://doi.org/10.1038/s41467-018-03296-8.
Wagner, A. 1862. “On a New Fossil Reptile Supposed to be Furnished with Feathers.” Annals and Magazine of Natural History, Third series, 9: 261–267.
Wang Yan, Han Hu, Jingmai K. O’Connor, Min Wang, Xing Xu, Zhonghe Zhou, Xiaoli Wang, and Xiaoting Zheng. 2017. “A Previously Undescribed Specimen Reveals New Information on the Dentition of Sapeornis chaoyangensis.” Cretaceous Research 74 (June): 1–10.
Wedel, Mathew J. 2006. “Origin of Postcranial Skeletal Pneumaticity in Dinosaurs.” Integrative Zoology 2: 80–85. https://doi.org/10.1111/j.1749-4877.2006.00019.x.
Wedel, Mathew. 2007. “What Pneumaticity Tells Us About ’Prosauropods’, and Vice Versa.” Special Papers in Palaeontology 77: 207–222.
Wedel, Mathew John. 2009. “Evidence for Bird-Like Air Sacs in Saurischian Dinosaurs.” Journal of Experimental Zoology 311A, no. 8 (1 October): 611–628.
Weishampel, David B., Peter Dodson, and Halszka Osmólska. 2004. The Dinosauria. 2nd ed. Berkeley, California: University of California Press.
Wellnhofer, P. 1993. “Das siebte Exemplar von Archaeopteryx aus den Solnhofener Schichten.” Archaeopteryx 11: 1–47.
Wellnhofer, Peter. 2009. Archaeopteryx: The Icon of Evolution. Munich, Germany: Dr. Friedrih Pfeil.
Woodward, Henry. 1862. “On a Feathered Fossil From the Lithographic Limestone of Solnhofen.” Intellectual Observer 2: 313–319.
Xu, Xing, Hailu You, Kai Du, and Fenglu Han. 2011. “An Archaeopteryx-like Theropod from China and the Origin of Avialae.” Nature 475, no. 7357 (27 July): 465–470.
Xu, Xing, and Diego Pol. 2014. “Archaeopteryx, Paravian Phylogenetic Analyses, and the Use of Probability-Based Methods for Palaeontological Datasets.” Journal of Systematic Palaeontology 12, no. 3 (May): 323–334.
Zhou, Zhonghe, Julia Clarke, and Fucheng Zhang. 2008. “Insight into Diversity, Body Size, and Morphological Evolution From the Largest Early Cretaceous Enantiornithine Bird.” Journal of Anatomy 212, no. 5 (May): 565–577.
















