The views expressed in this paper are those of the writer(s) and are not necessarily those of the ARJ Editor or Answers in Genesis.
Abstract
In a recent paper, O’Micks analyzed the cranial and postcranial characters for six hominin taxa and concluded that the recently discovered Homo naledi was “probably not part of the human holobaramin.” Consideration of postcranial characters is desirable in baraminology; however, previous studies of hominins have shown that reducing the taxon sample size can result in spurious baraminic distance correlations not seen in larger taxon samples. Re-analysis of O’Micks’ character sets show that the lack of baraminic distance correlations is likely the result of a small taxon sample. Therefore, excluding Homo naledi from the human holobaramin is an unwarranted conclusion from O’Micks’ results.
Keywords: Homo naledi, statistical baraminology, hominin, hominid.
Distinguishing human beings from animals based on a fragmentary fossil record remains a challenging and somewhat controversial problem among young-age creationists. Whereas statistical baraminology can offer some guidance in recognizing members of a baramin (e.g., Wood 2016a), the dearth of fossils, taxa, and character states prove challenging to the methodology. Despite the many fossils discovered in the last 20 years (Wood 2016b), even evolutionists are uncertain of how to name individual species (e.g., see Wood and Boyle 2016). Of particular interest to creationists are the hominin baraminology studies that focus almost exclusively on craniodental characteristics (O’Micks 2016a; Wood 2010, 2016b), which falls short of the ideal of the “holistic” character sets recommended by Wood and Murray (2003, 24–29). Since postcranial characteristics have traditionally been relied upon to distinguish human from nonhuman fossils (e.g., Hartwig-Scherer 1998), integrating postcranial and cranial characters for statistical baraminology of hominins should be a priority for creationists interested in hominin baraminology.
Given the importance of postcranial characters to hominin baraminology, O’Micks’ (2016b) novel analysis of the postcranial characters of Homo sapiens, H. neanderthalensis, H. naledi, Australopithecus sediba, A. africanus, and A. afarensis is a welcome contribution to a creationist understanding of the hominin fossil record. O’Micks also introduced a novel weighting scheme to give 62 craniodental and 37 postcranial characters equal weight when calculating baraminic distances. Although baraminic distances are traditionally calculated in an unweighted scheme to avoid researcher bias, the weighting scheme seems justified in this instance, given the importance of postcranial characters. Based on his analysis, O’Micks concluded that H. naledi and A. sediba probably do not belong to the human holobaramin.
O’Micks’ attempt to integrate postcranial characters into hominin baraminology closely resembles a previous attempt by Wood (2013) to use postcranial characters to resolve the relationship of A. sediba to the human holobaramin. In both cases, the addition of postcranial characteristics to an otherwise craniodental character matrix necessitated reducing the taxon sample size to a small fraction of the full set of taxa known from craniodental remains. In Wood’s composite postcranial and craniodental character matrices, the taxon sample size was reduced to five and six, depending on the source of postcranial characters. O’Micks used a similar taxon sample size of just six taxa. Wood observed poor baraminic distance correlation (BDC) after integrating postcranial characteristics, as did O’Micks. In contrast, Wood also evaluated exclusively craniodental characters using the much smaller taxon sample size and found poor BDC just as observed with the composite cranial/postcranial characters. Thus, the poor BDC was an artifact of small taxon sample size. Whereas O’Micks concluded that his analysis supported excluding Homo naledi from the human holobaramin, Wood (2013) concluded that the BDC results only revealed the importance of a large taxon sample to the calculation of BDC and added little to our understanding of the human holobaramin.
Here, BDC is calculated for three different sets of characters using the same six taxa used by O’Micks (2016b) using standard unweighted baraminic distances. All calculations filtered characters at a character relevance of 0.75, and bootstrap values were obtained from 100 pseudo-replicates of each character set. The first set of characters are O’Micks (2016b) 37 postcranial characters. After filtering for character relevance, 26 characters were used to calculate baraminic distance and BDC. The results (Fig. 1A) reveal only two instances of significant, positive BDC, between H. sapiens and H. neanderthalensis and between H. naledi and A. africanus. Bootstrap values for these positive correlations exceeded 90%. There were also five instances of significant, negative BDC between H. sapiens, H. neanderthalensis and other taxa, but these negative correlations had poor bootstrap values (<90%).
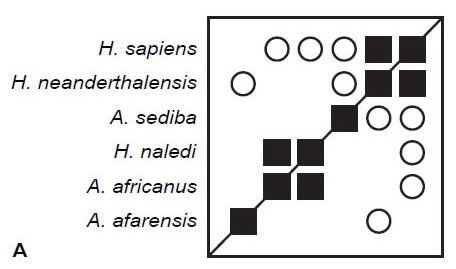
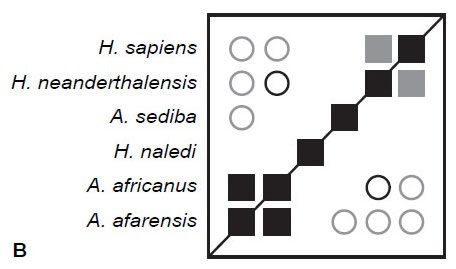
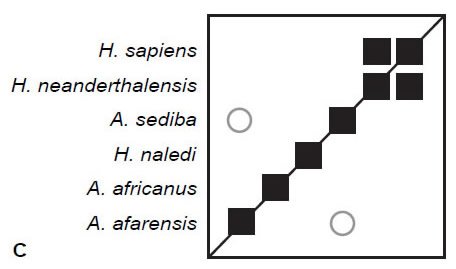
Fig. 1. BDC results for O’Micks’ (2016b) postcranial characters (A), O’Micks’ craniodental characters (B), and Berger et al.’s craniodental characters (C). Squares indicate taxa with significant, positive BDC; circles indicate taxa with significant, negative BDC. Black symbols have bootstrap values (100 replicates) >90%; gray symbols have bootstrap values ≤90%.
The second set of characters are O’Micks’ (2016b) 62 cranial characters. After filtering for character relevance, 55 characters were used to calculate baraminic distance and BDC. Again, the results (Fig. 1B) reveal two instances of significant, positive BDC, between H. sapiens and H. neanderthalensis (bootstrap value 88%) and between A. afarensis and A. africanus (bootstrap value 94%). Significant, negative BDC was observed for five taxon pairs, all involving A. africanus or A. afarensis, but only one taxon pair (H. neanderthalensis and A. africanus) exhibited both significant, negative BDC and a high bootstrap value (97%).
Finally, the 87 craniodental characters published by Berger et al. (2015) in Supplemental Table 2 were analyzed. After filtering for character relevance, all 87 characters were used to calculate baraminic distances and BDC. The results (Fig. 1C) reveal only two significant correlations: positive BDC with a 91% bootstrap value between H. sapiens and H. neanderthalensis and negative BDC with a 51% bootstrap value between H. naledi and A. afarensis.
Multidimensional scaling of each set of baraminic distances does not add substantively to the interpretation of the BDC results. All three datasets show diffuse clouds of taxa with little clustering (Fig. 2), as seen in Wood’s (2013) results.
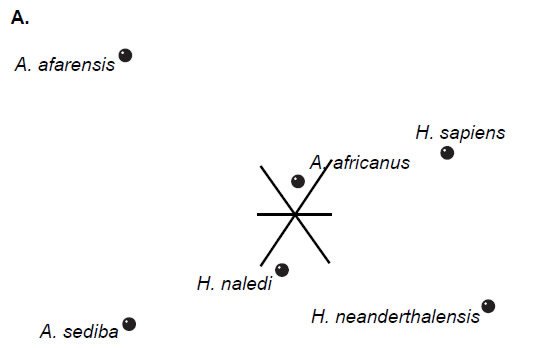
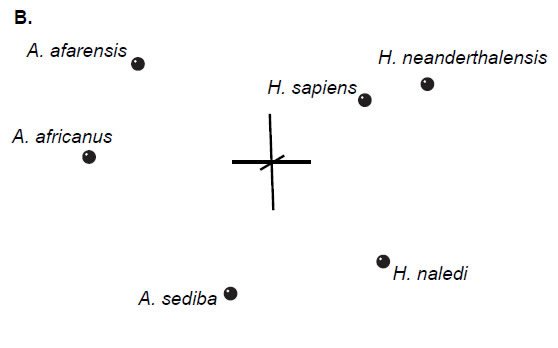
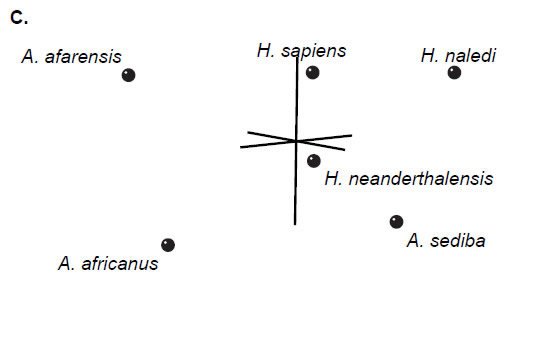
Fig. 2. Three-dimensional MDS results for O’Micks’ (2016b) postcranial characters (A), O’Micks’ craniodental characters (B), and Berger et al.’s craniodental characters (C). MDS for O’Micks’ postcranial characters had a 3D and minimal stress of 0.07. MDS for O’Micks’ craniodental characters had a 3D and minimal stress of 0.07. MDS for Berger et al.’s craniodental characters had a 3D and minimal stress of 0.05.
In each of these character sets, we see sparse BDC with poor bootstrap values, which parallels O’Micks’ (2016b) results based on a composite character set with weighted baraminic distances. Most importantly, we find here that reducing the taxon sample for craniodental characters results in a loss of BDC that was observed in the larger sample, as Wood (2013) observed previously. Wood’s (2016b) analysis of Berger et al.’s (2015) 87 craniodental characters revealed significant, positive BDC between all taxon pairs within genus Homo (Fig. 3). Here, with the same 87 characters, reducing the sample size to just six taxa results in only one correlation within genus Homo. Thus, the loss of statistically significant BDC reflects a statistical artifact arising from a small taxon sample size.
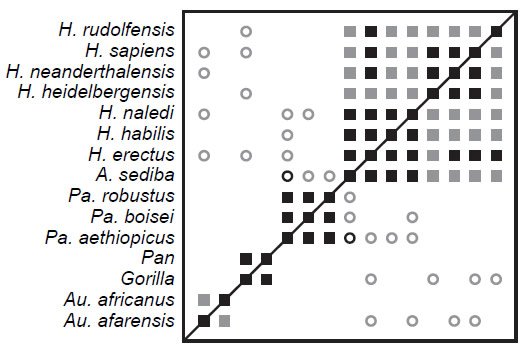
Fig. 3. BDC results for the craniodental characters and full taxon sample of Berger et al. (2015), after Wood’s (2016b) Figure 7. Squares indicate taxa with significant, positive BDC; circles indicate taxa with significant, negative BDC. Black symbols have bootstrap values (100 replicates) >90%; gray symbols have bootstrap values ≤90%.
We see a similar loss of significant BDC when partitioning O’Micks’ (2016b) characters into craniodental characters and postcranial characters. In each instance, we find sporadic BDC exactly as observed in O’Micks’ weighted analysis of the composite character set. More importantly, BDC observed in the present analyses and in O’Micks’ (2016b) composite set differ substantially from O’Micks’ (2016a) original analysis of just craniodental characters using larger taxon samples (Fig. 4). O’Micks originally found no significant, positive BDC between H. sapiens and H. neanderthalensis, which was found in the reduced taxon sample here. Likewise, H. naledi and H. neanderthalensis exhibited significant, positive BDC with the larger taxon sample size but not with the smaller taxon sample size.
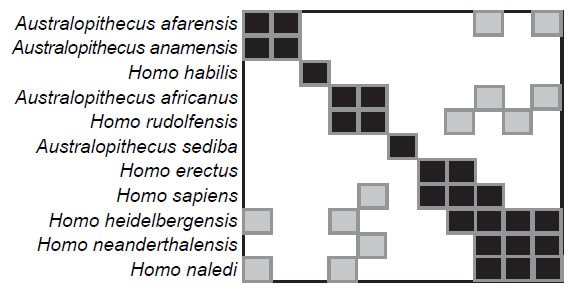
Fig. 4. BDC results for the craniodental characters and full taxon sample of O’Micks, from O’Micks’ (2016b) Fig. 1. Dark squares indicate taxa with significant, positive BDC; light squares indicate taxa with significant, negative BDC.
Thus, an evaluation of the effect of taxon sample size on O’Micks’ (2016b) results indicates that the observed change in correlations cannot be unambiguously attributed to the addition of new characters rather than the reduced taxon sample size. Further, even if we concede that were biologically relevant, the lack of significant positive or negative correlations for any taxon pair involving H. naledi (Fig. 4) should be interpreted as a lack of evidence regarding H. naledi’s baraminic affinities. Lack of correlation could be the result of statistical artifacts (as shown here), discontinuity, or extreme intrabaraminic diversification. O’Micks’ (2016b) results cannot distinguish these possible explanations, and here we see that his results are likely statistical artifacts.
What should we say then about Homo naledi? The current statistical baraminology results (O’Micks 2016a, b; Wood 2016b) all support either including H. naledi in the human holobaramin or at the very least not excluding it from the human holobaramin. The evidence of intentional burial or “body disposal” (Dirks et al. 2015; Randolph-Quinney et al. 2016) also supports recognizing Homo naledi as human (Wise 2016). This certainly challenges our understanding of human postcranial diversity, but there is no a priori reason to believe that humans could not diversify in body form. We already know that human postcranial diversity was once greater than it is today, since the Old Testament records the existence of giants (e.g., Deuteronomy 3:11, 2 Samuel 21:15–22), which must have had a much larger postcranial skeleton than extant humans. Most importantly, we must remember that all human beings, whatever their appearance, are descendants of Adam and Eve, made in the image of God, and eligible for salvation by faith in Christ’s redemptive work on the cross.
References
Berger, L. R., J. Hawks, D. J. de Ruiter, S. E. Churchill, P. Schmid, L. K. Delezene, T. L. Kivell, et al. 2015. “Homo naledi, a New Species of the Genus Homo from the Dinaledi Chamber, South Africa.” eLife 4: e09560.
Dirks, P. H. G. M., L. R. Berger, E. M. Roberts, J. D. Kramers, J. Hawks, P. S. Randolph-Quinney, M. Elliott, et al. 2015. “Geological and Taphonomic Context for the New Hominin Species Homo naledi From the Dinaledi Chamber, South Africa.” eLife 4: e09561.
Hartwig-Scherer, S. 1998. “Apes or Ancestors? Interpretations of the Hominid Fossil Record Within Evolutionary and Basic Type Biology.” In Mere Creation. Edited by W. A. Dembski, 212–235. Downers Grove, Illinois: InterVarsity Press.
O’Micks, J. 2016a. “Preliminary Baraminological Analysis of Homo naledi and its Place within the Human Baramin.” Journal of Creation Theology and Science Series B: Life Sciences 6: 31–39.
O’Micks, J. 2016b. “Homo naledi Probably Not Part of the Human Holobaramin Based on Baraminic Re-Analysis Including Postcranial Evidence.” Answers Research Journal 9: 263–272.
Randolph-Quinney, P. S., L. R. Backwell, L. R. Berger, J. Hawks, P. H. G. M. Dirks, E. M. Robergs, G. Nhauro, and J. Kramers. 2016. “Response to Thackeray (2016)—The Possibility of Lichen Growth on Bones of Homo naledi: Were They Exposed to Light? South African Journal of Science 112 (9/10): 1–5.
Wise, K. P. 2016. “Paleontological Note on Homo naledi.” Journal of Creation Theology and Science Series B: Life Sciences 6: 9–13.
Wood, B., and E. K. Boyle. 2016. “Hominin Taxic Diversity: Fact or Fantasy?” American Journal of Physical Anthropology 159: S37–S78.
Wood, T. C. 2010. “Baraminological Analysis Places Homo habilis, Homo rudolfensis, and Australopithecus sediba in the Human Holobaramin.” Answers Research Journal 3: 71–90.
Wood, T. C. 2013. “Australopithecus sediba, Statistical Baraminology, and Challenges to Identifying the Human Holobaramin.” In Proceedings of the Seventh International Conference on Creationism. Edited by M. Horstemeyer. Pittsburgh, Pennsylvania: Creation Science Fellowship.
Wood, T. C. 2016a. “A List and Bibliography of Identified Baramins.” Journal of Creation Theology and Science Series B: Life Sciences 6: 91–101.
Wood, T. C. 2016b. “An Evaluation of Homo naledi and “Early” Homo from a Young-Age Creationist Perspective.” Journal of Creation Theology and Science Series B: Life Sciences 6: 14–30.
Wood, T. C., and M. J. Murray 2003. Understanding the Pattern of Life: Origins and Organization of the Species. Nashville, Tennessee: Broadman & Holman.