The views expressed in this paper are those of the writer(s) and are not necessarily those of the ARJ Editor or Answers in Genesis.
Abstract
A review of vestigial claims of the human spleen reports no clear evidence for its function until the late 1950s. It is now acknowledged to be a critical organ serving at least six different important functions. Furthermore, the spleen exists universally in jawed vertebrates and is not found in any invertebrates, so it has no evolutionary history. The lack of evidence for simpler or transitional forms of the spleen supports the conclusion that jawed vertebrates have always had fully functional spleens that are perfectly designed for their needs.
Keywords: Spleen, vestigial organs, human body, lymphatic system, Intelligent Design, Darwinism, evolution of body organs
Introduction
Claims that progress in the field of anatomy and medicine has been impeded by Darwin’s idea of useless organs that he called rudimentary organs. Sarker et al. opined:
According to the evolutionary knowledge of comparative anatomy, many organs in the human body can be categorized as vestiges. Vestigial in that sense that the organ seemingly has no functions or useless now but they might have functions previously which are lost someway due to evolution. (Sarker et al. 2015, 11)
Sarker concluded that before an organ is labeled vestigial careful evaluation is required to determine if it has a function. As one review correctly noted, the Appendix, tonsils, various redundant veins—they’re all vestigial body parts once considered expendable, if not downright useless. But as technology has advanced, researchers have found that, more often than not, some of these “junk parts” are actually hard at work. (Koerth-Baker 2009)
One example that illustrates this trend is the spleen, an organ which research has now proven to be critical to good health. The spleen is part of the lymphatic system that, with rare exceptions, exists in all vertebrates (Marieb and Hoehn 2017, 667). As a result of damage from sports injuries, automobile accidents or disease, it sometimes must be surgically removed (Martin 1995, 790). Overall, with proper remedial care, most splenectomy patients can usually live a fairly normal life. For this reason, “for most of medical history, the spleen was considered nonessential to life.” (Nathan 1981, 888).
In his classic documentation of the close-to-100-claimed human vestigial organs, Wiedersheim included the spleen because, in the “placental mammals the lobes are increasingly reduced” and “in the primates, the posterior lobe [of the spleen] has almost disappeared” showing, he argued, as we go up the evolutionary ladder from the lower to the higher animals, the spleen is gradually disappearing (Wiedersheim 1895, 186). In man, Wiedersheim claimed, the posterior lobe of the spleen has almost totally disappeared due to evolution. Many early anatomy authorities, such as Wilder, mention the spleen, but do not discuss its function (Wilder 1909, 293, 363). In contrast, some modern authorities at least speculate on its possible functions (Schmidt-Rhaesa 2007). Likely the reason is because, until recently, it was considered nonessential (Schiller 1991, 205). Some early anatomists, though, recognized hints of the spleen’s actual functions. Kingsley noted “Recent studies tend to show that the spleen may play an important part in the formation of anti-bodies and thus has a value in resistance to infection. It may also have an indirect connection with the formation of erythrocytes” (Kingsley 1926, 348).
Description of the Spleen
The human spleen is a fist-shaped, purple-colored organ about 10 cm (4 in) long located on the upper-left side of the abdomen behind the stomach. The splenic artery carries blood into the spleen, and the processed blood exits by the splenic vein. The healthy adult spleen weighs around 200 g but certain medical conditions can cause splenomegaly, an enlargement that indicates great strain on the spleen. An enlarged spleen results from attempts to deal with infections, such as malaria, liver disease and certain blood cancers (Schiller 1991). Glandular fever (infectious mononucleosis) can also enlarge the spleen, and, if extreme, can lead to spontaneous rupture.
The spleen is composed of a reticular connective tissue framework surrounded by a fibrous connective tissue capsule. Inside the capsule is a complex network of vessels and compartments separated by discrete trabecular partitions (fig. 1). The interior of the spleen contains two tissue types, white pulp and red pulp. The white pulp contains clusters of white corpuscles containing lymphocytes and macrophages and the red pulp has thick masses of erythrocytes (Bowdler 2002, 12,13). Because spleens contain large amounts of blood, they are relatively vulnerable to injury.
Although the spleen is effectively protected by the rib cage, injuries can be serious. Fractured ribs, such as from a car accident or playing football, can puncture the spleen. Because the spleen is highly vascular, this puncture can cause bleeding out into the abdomen, resulting in large amounts of blood leaving the circulating bloodstream. This bleeding can be life-threatening, and, consequently, surgery to remove the spleen (splenectomy) is often done in this emergency.
When the spleen is injured, cells from the damaged organ can scatter throughout the abdomen. If the cells land in locations with many blood vessels, they can begin to grow into tiny extra spleens called splenunculi (a process called splenosis). The functional importance of such splenuculi is uncertain (Rashid 2014).
If the spleen is removed surgically, another organ (often the liver) will usually take over some or many of its functions (Tischendorf 1985, 145). Nonetheless, for reasons not yet fully understood, a splenectomy drastically shortens the lifespan in a small number of patients. In most of these particular patients, all of its major functions cannot be assumed by another organ after the spleen has been removed (Bowdler 2002).
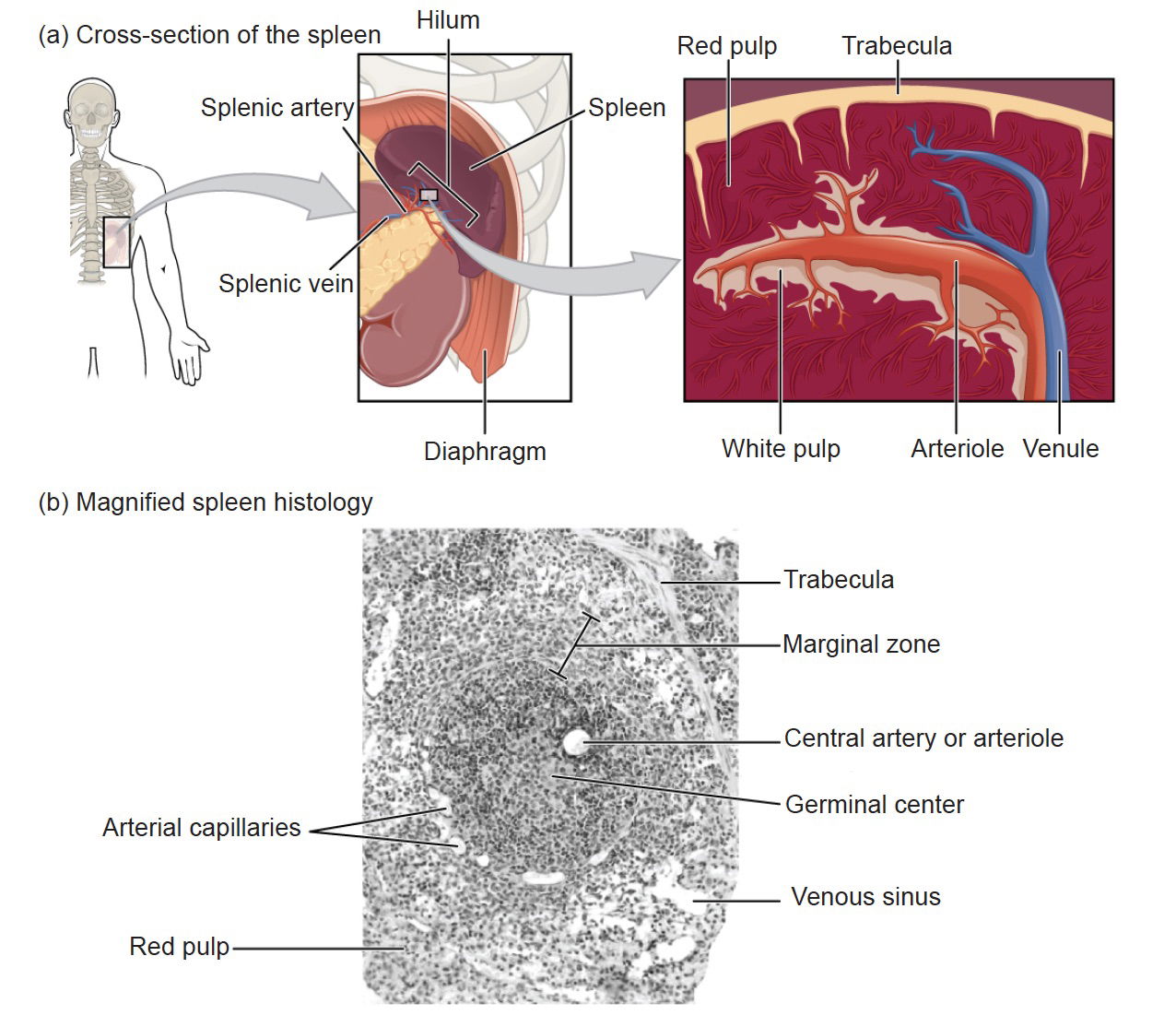
Fig. 1. The location and details of the spleen. Image courtesy of Wikimedia Commons. https://upload.wikimedia.org/wikipedia/commons/f/ff/2208_Spleen.jpg
Functions of the Spleen
The functions of the spleen primarily relate to circulation (maintaining blood cells and blood volume) and immunity (filtering blood and housing immune cells). One of the earliest functions of the spleen is in the fetus, where it manufactures red blood cells and blood platelets used for blood clotting. One reason the spleen was labeled vestigial by some Darwinists, at least for red corpuscle production, is because it ordinarily does not fulfill these blood-forming functions in adults (King and Showers 1964, 303). Recently it has been documented that, in cases of severe hemorrhage or extreme anemia, the spleen actually resumes the function of blood cell synthesis until the emergency has passed.
The spleen also participates in the removal of red blood cells and blood platelets that are damaged, defective, and worn-out. The average lifespan of red blood cells is about 120 days. They then must be broken down and the parts recycled or disposed; this job is primarily performed by macrophages in the spleen. The spleen can also help stabilize blood volume after hemorrhagic shock causes large blood losses, for example in a bad car accident, by releasing blood back into the circulatory system.
One of the spleen’s major known functions is to help to locate and fight infections. The spleen’s evaluation strategies involves blood slowing down as it passes through the spleen, giving the immune system more time to recognize and produce antibodies (Hammerquist et al. 2016). This process is an important part of an effective immune response that can kill bacteria before the infection is noticeable. If a person has had their spleen removed, is born without a spleen (asplenia), or diagnosed with a low-functioning spleen (hyposplenism), they have a significant increased lifelong risk of bacterial infections. The most common bacteria that cause infections in asplenic people are Pneumococcus, Meningococcus, and Haemophilus influenzae type B (Hammerquist et al. 2016, 220). These bacteria are transferred from person to person through saliva or mucus droplets from a cough or sneeze. In healthy persons, this type of bacteria rarely causes illness.
The spleen is uniquely designed to identify “stealthy” bacteria that are encapsulated by a difficult to detect polysaccharide coating. Once a patient has lost their spleen, it is recommended that they receive immunizations for these encapsulated bacteria, some of which were named above.
Research on mice has discovered that the spleen stores monocytes, a type of white blood cell essential both for immune defense and tissue repair. This is especially important in microbial infections, myocardial infarctions, or gaping wounds. Monocytes are produced in red bone marrow, and they then travel through the bloodstream to where they are needed in the body. New research has found evidence that the spleen contains ten times as many monocytes as the blood, thus is a far more important monocyte storehouse than previously thought. Monocytes are recruited and sent to tissue injury sites and also trigger inflammation to help control the infection. Monocytes are also important to aid in tissue repair. The researchers concluded that “bona fide undifferentiated monocytes reside in the spleen and outnumber their equivalents in circulation. The reservoir monocytes assemble in clusters in the cords of the subcapsular red pulp and are distinct from macrophages and DCs (dendritic cells).” They added that, in response to ischemic myocardial injury, splenic monocytes increase their motility, exit the spleen en masse, accumulate in injured tissue, and participate in wound healing. These observations uncover a role for the spleen as a site for storage and rapid deployment of monocytes and identify splenic monocytes as a resource that the body exploits to regulate inflammation (Swirski et al. 2009, 612).
Surviving a heart attack requires healing that depends on monocytes, thus the critical role of the spleen in heart attack recovery. The researchers also found from evaluations of lab mice that the spleen is the source of close to half of the monocytes involved recovery after a heart attack.
Swirski, the lead author of one spleen study, wrote: “It was thought that the monocytes that accumulated immediately after a heart attack were ones that had been circulating in the blood. But we did calculations and found that the number that accumulated in the heart far exceeded the number in circulation . . . in studies where we removed the spleen and then induced a heart attack, we saw a vastly fewer number of monocytes accumulate” (quoted in Koerth-Baker 2009).
In short, the mice without spleens were not able to recover from the heart attack nearly as well, if at all. Now we understand that humans who lack a functional spleen likewise do not recover nearly as well.
A 1977 study published in the medical journal, The Lancet, followed the health of 740 World War II veterans over 20 years. The study compared veterans with and without spleens, mostly spleens lost due to war injuries. The spleenless men were twice as likely to die from ischemic heart disease and/or pneumonia and were also slightly more likely to die of liver cirrhosis (Robinette and Fraumeni 1977, 127). One reason they were more likely to die of pneumonia is that they lacked the ability to identify, and to destroy, the encapsulated bacterium Pneumococcus.
Other Functions of the Spleen
Since the spleen houses large numbers of white blood cells, it is also an important source of immune proteins—such as opsonins, properdin convertase, and tuftsin—produced by those cells. Opsonins are molecules that enhance phagocytosis by marking antigens on enemy cells to increase the body’s protective immune response; they also mark dead cells for recycling, thereby helping to remove detritus from the body. Properdin convertase, or Factor P, is a protein family that activates other proteins to carry out their role of protecting the body, while tuftsin is a tetra-peptide that has an immune-stimulatory effect.
These systems are all critical to overall health, and consequently those who lack a functional spleen are more likely to die of, not only heart disease, but also pneumonia and other diseases. Professor Swirski noted that we knew the spleen played an important role in the body, but we didn’t know specifically how it accomplished its wonders. Now we know how (Koerth-Baker 2009).
Useless Organs Is Dangerous Logic
Dr. Jeffrey Laitman, Director of the Department of Anatomy and Functional Morphology at the Mount Sinai School of Medicine observed that the vestigial organ idea birthed by evolution is dangerous both to health and knowledge because history is littered with body parts that were called “useless” simply because medical science had yet to understand them. People say, You can remove it and still live. But you have to be careful with that logic. You could remove your left leg and still live. But whenever a body part is moved or changed, there’s a price to pay. (Koerth-Baker 2009)
This is very wise advice, as the history of our understanding of the spleen reviewed above has documented. The spleen has moved from a useless organ that is gradually shrinking as we go up the purported evolutionary ladder from lower to higher animals, to a critical player in maintaining good health and helping our body heal.
The Spleen’s Presence in Animals
Although its morphology and physiology varies in different animals, the spleen is found in all jawed vertebrates and in no other animal. In most vertebrates, the spleen continues to produce red blood cells throughout life; only in mammals is this function lost in middle-aged adults. In cartilaginous and ray-finned fish, the spleen consists primarily of red pulp. It is somewhat elongated in ray-finned fish because it is located inside the intestinal serosal lining. Reptiles, birds and mammals contain large amounts of white pulp. In many amphibians, such as frogs, it assumes a more rounded form and often a greater quantity of white pulp (Romer and Parsons 1986, 450–451).
Birds and mammals typically have a round spleen, adjusted according to its surrounding organs. Many mammals have tiny spleen-like structures known as haemal nodes throughout the body that are presumed to have the same function as the spleen. The aquatic mammalian spleen differs in minor ways from those of land-dwelling mammals, such as in general they are bluish in color. In cetaceans and manatees, the spleens tend to be very small, but in deep diving pinnipeds, the spleen is massive in order to store large numbers of red blood cells that allow them to survive their long stays under water. Furthermore, no evidence exists of a splenic evolutionary transition between vertebrates and invertebrates. Vertebrates that are less hygienic, like dogs and cats, have spleens that are proportionately much larger than human spleens. This alteration by our Creator enables scavengers to consume bacteria infested food sources and, because of the added immune surveillance, to evade infection.
Summary
The spleen, once regarded as a useless vestige by many evolutionists, has now been documented to have numerous important roles in the body, including recycling red blood cells, filtering blood, and housing immune cells such as lymphocytes and monocytes as well as serving as a reservoir for monocytes or plasma that may be needed elsewhere. The useless organ idea has impeded looking for the actual function of numerous organs and structures, and the spleen is just one more example. All of the formerly claimed vestigial organs have now been confirmed to have one or more functions (Bergman 2019).
The spleen is a critical organ in all jawed vertebrates and is not present in any invertebrate. Furthermore, no evidence exists for its evolution from a non-spleen. The available evidence suggests that the earliest jawed vertebrates had functional spleens identical to those existing in their modern counterparts. Research continues to reveal, “the spleen is an amazing organ that clearly shows the wisdom and handiwork of our creator” (Menton 2011, 69).
References
Bergman, Jerry. 2019. Useless Organs: The Rise and Fall of the Once Major Argument for Evolution. Tulsa, Oklahoma: Bartlett Publishing.
Bowdler, Anthony. 2002. The Complete Spleen: Structure, Function, and Clinical Disorders. 2nd edition. Totowa, New Jersey: Humana Press.
Hammerquist, Rhonda J., Kimberly A. Messerschmidt, April A. Pottebaum, and Thaddaus R. Hellwig. 2016. “Vaccinations in Asplenic Adults.” American Journal of Health-System Pharmacy 73, no. 9 (May): e220–e228. https://doi.org/10.2146/ajhp150270.
King, Barry G. and Mary J. Showers. 1964. Human Anatomy and Physiology. Philadelphia, Pennsylvania: W. B. Sanders.
Kingsley, John Sterling. 1926. Comparative Anatomy of Vertebrates. 3rd ed. (Revised). Philadelphia, Pennsylvania: P. Blakiston’s Son & Co.
Koerth-Baker, Maggie. 2009. “Vestigial Organs Not So Useless After All, Studies Find.” National Geographic News, July 30.
Marieb, Elaine N., and Katja Hoehn. 2017. Anatomy and Physiology. San Francisco, California: Pearson Publishing Company.
Martin, Frederic. 1995. Fundamentals of Anatomy and Physiology. Englewood Cliffs, New Jersey: Prentice Hall.
Menton, David. 2011. “The Mysterious Spleen.” Answers Research Journal 6, no. 4 (October 1): 68–71. https://answersingenesis.org/human-body/the-mysterious-spleen/.
Nathan, David, and Frank Oski. 1981. Hematology of Infancy and Childhood. Vol. 2. Philadelphia, Pennsylvania: Saunders.
Rashid, Sameeah Abdulrahman. 2014. Accessory Spleen: Prevalence and Multidetector CT Appearance. Malay Journal of Medical Science21, no. 4 (July): 18–23.
Robinette, C. Dennis, and Joseph F. Fraumeni Jr. 1977. “Splenectomy and Subsequent Mortality in Veterans of the 1939–45 War.” The Lancet 310, no. 8029 (July 16): 127–129.
Romer, Alfred Sherwood, and Thomas S. Parsons. 1986. The Vertebrate Body. Philadelphia, Pennsylvania: Holt- Saunders International.
Sarker, Aniruddha, Anubha Saha, Sanchita Roy, Santanu Pathak, and Shyamash Mandal. 2015. “A Glimpse Towards the Vestigiality and Fate of Human Vermiform Appendix—A Histomorphometric Study.” Journal of Clinical and Diagnostic Research. 9(2): (February) AC11- AC15.
Schiller, Medad. 1991. Pediatric Surgery of the Liver, Pancreas, and Spleen. Philadelphia, Pennsylvania: Sanders.
Schmidt-Rhaesa, Andreas. 2007. Evolution of Organ Systems. New York, New York: Oxford University Press.
Swirski, Filip K., Matthias Nahrendorf, Martin Etzrodt, Moritz Wildgruber, Virna Cortez-Retamozo, Peter Panizzi, Jose-Luiz Figueiredo, et al. 2009. “Identification of Splenic Reservoir Monocytes and Their Deployment to Inflammatory Sites.” Science 325, no. 5940 (July 31): 612– 616.
Tischendorf, F. 1985. “On the Evolution of the Spleen.” Experientia 41, no. 2 (February): 145–152.
Wiedersheim, Robert. 1895. The Structure of Man: An Index to His Past History. Translated by H. M. Bernard. London, United Kingdom: Macmillan and Co.
Wilder, Harris Hawthorne. 1909. The Story of the Human Body. New York, New York: Henry Holt and Company.